
Concept explainers
An acid-base reaction of
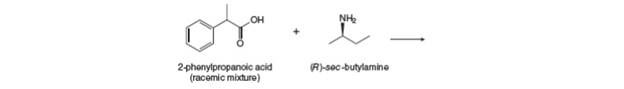
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: Atoms First
General Chemistry: Atoms First
Chemistry by OpenStax (2015-05-04)
General, Organic, and Biological Chemistry (3rd Edition)
- Two diastereomeric sets of enantiomers, A/B and C/D, exist for 3-bromo-2-butanol. When enantiomer A or B is treated with HBr, only racemic 2,3-dibromobutane is formed; no meso isomer is formed. When enantiomer C or D is treated with HBr, only meso 2,3-dibromobutane is formed; no racemic 2,3-dibromobutane is formed. Account for these observations.arrow_forwardConsider the stereochemistry of the equimolar mixture of compounds and its optical activity. Flm H3C F Which statement is true? Select one: the mixture is optically active the optical activity of the mixture cannot be determined by the information presented the mixture is optically inactivearrow_forwardThe southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. H3C- OH ✔arrow_forward
- Isopulegol is formed as a single stereoisomer. Account for the fact that only a single stereoisomer is formedarrow_forwardFirst calculate r value of each then draw all possible structural and stereochemical isomers. Include enols and enamines and any e and z isomers. -C3H4O -C3H6O -C4H8O -C4H9Narrow_forwardIn the presence of heat or light, diazomethane is converted into a carbene that adds to alkenes. Draw the correct products for the following reaction. Please draw stereochemistry at all chiral centers ( 2 flat bonds, 1 dash and 1 wedge).arrow_forward
- Draw the structures of the following compound: 8-benzyl-6-(3-chloro-2-methylbutyl)-N-ethyl-9-fluoro-N,2,2-trimethyl-7-propoxy-5-decanaminearrow_forwardDraw and name all the stereoisomers of 2-bromo-3-iodopentane. Indicate which stereoisomers are enantiomers and which are diastereomers.arrow_forwardDraw the products formed when (S)-butan-2-ol is treated with TsCl and pyridine, followed by NaOH. Label the stereogenic center in each compound as R or S. What is the stereochemical relationship between the starting alcohol and the nal product?arrow_forward
- Consider the reaction of (E)-hex-3-ene with HBr. Draw the structures for (E)-hex-3-ene and the two major organic products for its reversion with HBr. Show all stereoisomers using wedge and dash stereochemistry at any chirality centerarrow_forwardThe southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. HgC O OH aarrow_forwardDisregarding stereoisomers, how many different enols can the β-diketone CH3COCH2COCH2CH3 form?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

