
Concept explainers
Draw all possible stereoisomers for each cycloalkane. Label pairs of enantiomers and diastereomers. Label any meso compound.
a. 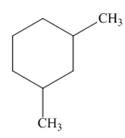 b.
b. 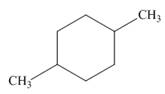 c.
c. 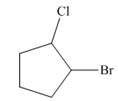
(a)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are three. Among the three stereoisomers, the labeling of two enantiomers of the given cycloalkane is
Explanation of Solution
The given cycloalkane is,
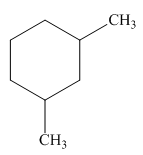
Figure 1
Due to the presence of cyclohexane ring attached with two methyl groups, the IUPAC name of this cycloalkane is
Where,
•
Substitute the number of chiral centers of
Thus, the total number of stereoisomers of
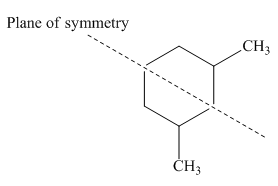
Figure 2
Thus, this compound forms only three stereoisomers instead of four. Among the three stereoisomers of
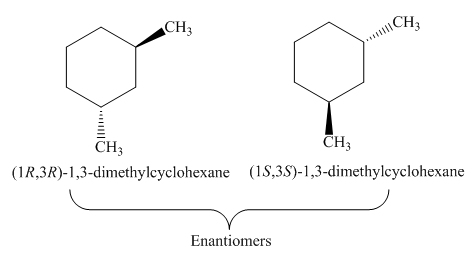
Figure 3
The remaining one stereoisomer of
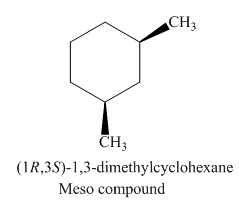
Figure 4
The possible stereoisomers for the given cycloalkane are three. Among the three stereoisomers, the labeling of two enantiomers of the given cycloalkane is
(b)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are two. Both are meso compounds and these meso compound are labeled as done as
Explanation of Solution
The given cycloalkane is,
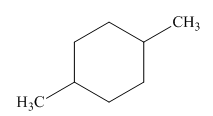
Figure 5
Due to the presence of cyclohexane ring attached with two methyl groups at first and fourth carbon atoms, the IUPAC name of this cycloalkane is
This
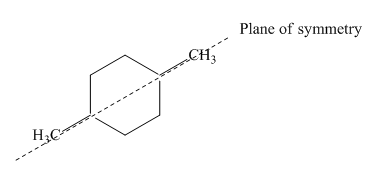
Figure 6
Thus, this compound forms only two stereoisomers one is cis isomer and second is trans isomer. The given compound,
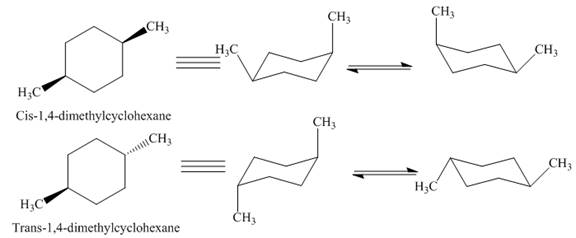
Figure 7
Thus,
The possible stereoisomers for the given cycloalkane are two. Both are meso compounds and these meso compound are labeled as done as
(c)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are four. It contains two pairs of enantiomers, the labeling of two pairs of enantiomers of the given cycloalkane is
Explanation of Solution
The given cycloalkane is,
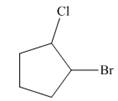
Figure 8
Due to the presence of cyclopentane ring attached with one bromine atom and one chlorine atom at first and second carbon atom respectively, the IUPAC name of this cycloalkane is
Where,
•
Substitute the number of chiral centers of
Thus, the total number of stereoisomers of
The four stereoisomers of
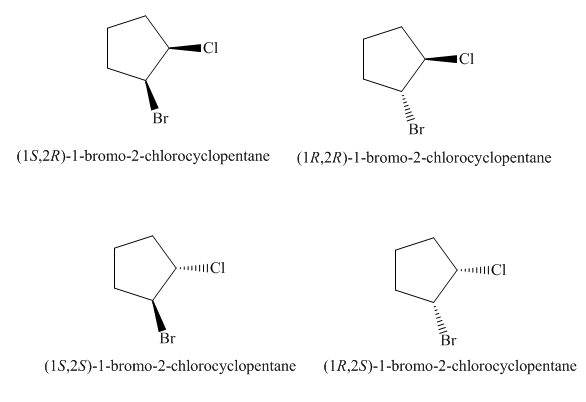
Figure 9
The compound,
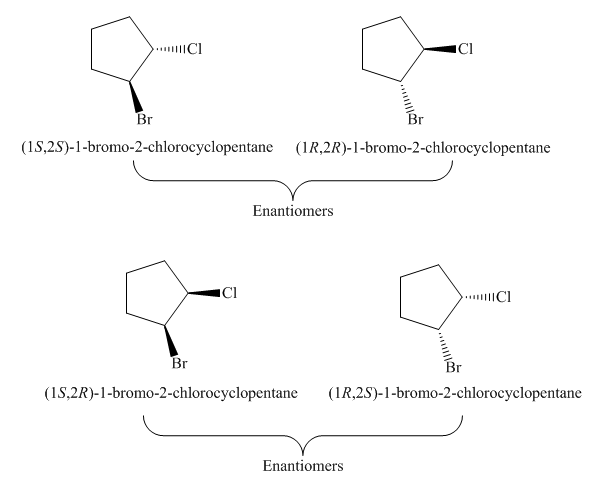
Figure 10
The possible stereoisomers for the given cycloalkane are four. It contains two pairs of enantiomers, the labeling of two pairs of enantiomers of the given cycloalkane is
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
- Neuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent.a.Label each carbon–carbon double bond as conjugated or isolated. b.Label each double bond as E or Z. c.For each conjugated system, label the given conformation as s-cis or s-trans.arrow_forward1. Draw the two conformations of trans-1-bromo-3-methylcyclohexane using both the Newman projection and the chair drawing. Calculate the energies of each. What ratio of the two conformers will be present at equilibrium? 2. Draw a structure of a chiral alcohol.arrow_forwardDrawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D? b.Label each compound as chiral or achiral. c.Which compounds alone would be optically active? d.Which compounds have a plane of symmetry? e.How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D? f.Which of the compounds are meso compounds? g.Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?arrow_forward
- • Question 5: Label each stereogenic center as R or S. CH₂ a. CH₂CH₂ b. CH H 1 NH₂ H CH3 CH₂CH₂ T C. D CI CH3 d. Br C ICH₂ H e. f. H* HO C HOOC CH₂ HO CH(CH3)2 -C₂ C CH3 SH NH₂ H CH3 g. h. .CI CIarrow_forwarda. Draw the two chair conformers for each of the stereoisomers of trans-1-tert-butyl-3-methylcyclohexane. b. For each pair, indicate which conformer is more stable.arrow_forwardDraw all alkenes that react with one equivalent of H2 in the presence of a palladium catalyst to form each alkane. Consider constitutional isomers only. a. b.arrow_forward
- Ethane spends all its time in the staggered conformation because it has the lowest potential energy. a. True b. False c. Neitherarrow_forwardConsider 1,2-dimethylcyclohexane. a.Draw structures for the cis and trans isomers using a hexagon for the sixmembered ring. b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable? c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable? d.Which isomer, cis or trans, is more stable and why?arrow_forward1. Rank the following conformers of the isomers of 1-ethyl-2,4-dimethylcyclohexane from most to least stable. JI. B. C. E D.arrow_forward
- Which of the following statements is not true regarding the conformation of substituted cyclohexanes? O a. the boat conformation of cyclohexane is usually more stable than the chair conformation O b. ring inversion of cyclohexane between two chair conformations takes place via a boat conformation O c. substituted cyclohexanes are destabilized by 1,3-diaxial interactions d. the relative amount of two conformations of substituted cyclohexanes can be determined from the difference in strain energyarrow_forwardDraw a chair conformation of cyclohexane with one CH3CH2 group and one CH3 group that fits each description. a.a 1,1-disubstituted cyclohexane with an axial CH3CH2 group b. a cis-1,2-disubstituted cyclohexane with an axial CH3 group c. a trans-1,3-disubstituted cyclohexane with an equatorial CH3 group d. a trans-1,4-disubstituted cyclohexane with an equatorial CH3CH2 group.arrow_forwardDrawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related (enantiomers, diastereomers,constitutional isomers): A and B; A and C; B and C; C and D?b. Label each compound as chiral or achiral.c. Which compounds, alone, would be optically active?d. Which compounds have a plane of symmetry?e. Which of the compounds are meso compounds?f. Would an equal mixture of compounds C and D be optically active? Whatabout an equal mixture of B and C?g. How many stereogenic centers are there for each compound?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





