
(a)
Interpretation:
The given triatomic molecule geometry has to be predicted using VSEPR theory.
Concept Introduction:
Information about the number of bonds and types of bonds can be obtained from Lewis structure but the molecular geometry cannot be obtained. Three dimensional arrangement of atoms in a molecule can be given by molecular geometry. Physical and chemical properties are determined by the molecular geometry of the molecule.
Using VSEPR theory and Lewis structure, the molecular geometry of the molecule that contain less number of atoms can be predicted. VSEPR theory uses the information from Lewis structure of the molecule to predict the molecular geometry of the molecule. Main concept of VSEPR theory is that electron pairs that are present in the valence shell adopt arrangement in a way that minimize the repulsion between like charges.
If the central atom contains two electron pairs, then it has to be far apart means, it has to be on opposite side of the nucleus. This means the angle has to be
If the central atom contains three electron pairs, then it has to be far apart means, it has to be on corner of a triangle. This means the angle has to be
If the central atom contains four electron pairs, then it has to be far apart means, it has to be in a tetrahedral arrangement. This means the angle has to be
The collection of valence electron that is present in localized region about central atom in a molecule is known as VSEPR electron group. This may contain two electrons, four electrons, or six electrons. The electron group that contain four and six electrons repel each other.
Tetrahedral VSEPR electron group:
The four electron pairs can be of three VSEPR electron groups. They are 4 bonding electron groups, 3 bonding and 1 nonbonding electron groups, and 2 bonding and 2 nonbonding electron groups. The molecular geometry that is associated with 4 bonding electron groups is tetrahedral. The molecular geometry that is associated with 3 bonding and 1 nonbonding electron groups is trigonal pyramidal. The molecular geometry that is associated with 2 bonding and 2 nonbonding electron groups is angular.
Trigonal planar VSEPR electron group:
The three electron pairs can be of two VSEPR electron groups. They are 3 bonding electron groups, and 2 bonding and 1 nonbonding electron groups. The molecular geometry that is associated with 3 bonding electron groups is trigonal planar. The molecular geometry that is associated with 2 bonding and 1 nonbonding electron groups is angular.
Linear VSEPR electron group:
The two electron pairs can be of only one VSEPR electron groups. It is only 2 bonding electron groups and the geometry associated with it is linear geometry.
(a)
Answer to Problem 5.49EP
The molecular geometry is angular (bent).
Explanation of Solution
Given triatomic molecule is,
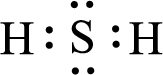
The central atom in the above molecule is found to be sulfur. This has two bonding electron groups and two nonbonding electron groups. The arrangement around the central atom is tetrahedral arrangement. Looking for molecular geometry, the central atom that contains two bonding electron groups and two nonbonding electron groups and it has angular geometry as per VSEPR theory.
Molecular geometry is predicted for the given triatomic molecule using VSEPR theory.
(b)
Interpretation:
The given triatomic molecule geometry has to be predicted using VSEPR theory.
Concept Introduction:
Information about the number of bonds and types of bonds can be obtained from Lewis structure but the molecular geometry cannot be obtained. Three dimensional arrangement of atoms in a molecule can be given by molecular geometry. Physical and chemical properties are determined by the molecular geometry of the molecule.
Using VSEPR theory and Lewis structure, the molecular geometry of the molecule that contain less number of atoms can be predicted. VSEPR theory uses the information from Lewis structure of the molecule to predict the molecular geometry of the molecule. Main concept of VSEPR theory is that electron pairs that are present in the valence shell adopt arrangement in a way that minimize the repulsion between like charges.
If the central atom contains two electron pairs, then it has to be far apart means, it has to be on opposite side of the nucleus. This means the angle has to be
If the central atom contains three electron pairs, then it has to be far apart means, it has to be on corner of a triangle. This means the angle has to be
If the central atom contains four electron pairs, then it has to be far apart means, it has to be in a tetrahedral arrangement. This means the angle has to be
The collection of valence electron that is present in localized region about central atom in a molecule is known as VSEPR electron group. This may contain two electrons, four electrons, or six electrons. The electron group that contain four and six electrons repel each other.
Tetrahedral VSEPR electron group:
The four electron pairs can be of three VSEPR electron groups. They are 4 bonding electron groups, 3 bonding and 1 nonbonding electron groups, and 2 bonding and 2 nonbonding electron groups. The molecular geometry that is associated with 4 bonding electron groups is tetrahedral. The molecular geometry that is associated with 3 bonding and 1 nonbonding electron groups is trigonal pyramidal. The molecular geometry that is associated with 2 bonding and 2 nonbonding electron groups is angular.
Trigonal planar VSEPR electron group:
The three electron pairs can be of two VSEPR electron groups. They are 3 bonding electron groups, and 2 bonding and 1 nonbonding electron groups. The molecular geometry that is associated with 3 bonding electron groups is trigonal planar. The molecular geometry that is associated with 2 bonding and 1 nonbonding electron groups is angular.
Linear VSEPR electron group:
The two electron pairs can be of only one VSEPR electron groups. It is only 2 bonding electron groups and the geometry associated with it is linear geometry.
(b)
Answer to Problem 5.49EP
The molecular geometry is angular (bent).
Explanation of Solution
Given triatomic molecule is,

The central atom in the above molecule is found to be oxygen. This has two bonding electron groups and two nonbonding electron groups. The arrangement around the central atom is tetrahedral arrangement. Looking for molecular geometry, the central atom that contains two bonding electron groups and two nonbonding electron groups and it has angular geometry as per VSEPR theory.
Molecular geometry is predicted for the given triatomic molecule using VSEPR theory.
(c)
Interpretation:
The given triatomic molecule geometry has to be predicted using VSEPR theory.
Concept Introduction:
Information about the number of bonds and types of bonds can be obtained from Lewis structure but the molecular geometry cannot be obtained. Three dimensional arrangement of atoms in a molecule can be given by molecular geometry. Physical and chemical properties are determined by the molecular geometry of the molecule.
Using VSEPR theory and Lewis structure, the molecular geometry of the molecule that contain less number of atoms can be predicted. VSEPR theory uses the information from Lewis structure of the molecule to predict the molecular geometry of the molecule. Main concept of VSEPR theory is that electron pairs that are present in the valence shell adopt arrangement in a way that minimize the repulsion between like charges.
If the central atom contains two electron pairs, then it has to be far apart means, it has to be on opposite side of the nucleus. This means the angle has to be
If the central atom contains three electron pairs, then it has to be far apart means, it has to be on corner of a triangle. This means the angle has to be
If the central atom contains four electron pairs, then it has to be far apart means, it has to be in a tetrahedral arrangement. This means the angle has to be
The collection of valence electron that is present in localized region about central atom in a molecule is known as VSEPR electron group. This may contain two electrons, four electrons, or six electrons. The electron group that contain four and six electrons repel each other.
Tetrahedral VSEPR electron group:
The four electron pairs can be of three VSEPR electron groups. They are 4 bonding electron groups, 3 bonding and 1 nonbonding electron groups, and 2 bonding and 2 nonbonding electron groups. The molecular geometry that is associated with 4 bonding electron groups is tetrahedral. The molecular geometry that is associated with 3 bonding and 1 nonbonding electron groups is trigonal pyramidal. The molecular geometry that is associated with 2 bonding and 2 nonbonding electron groups is angular.
Trigonal planar VSEPR electron group:
The three electron pairs can be of two VSEPR electron groups. They are 3 bonding electron groups, and 2 bonding and 1 nonbonding electron groups. The molecular geometry that is associated with 3 bonding electron groups is trigonal planar. The molecular geometry that is associated with 2 bonding and 1 nonbonding electron groups is angular.
Linear VSEPR electron group:
The two electron pairs can be of only one VSEPR electron groups. It is only 2 bonding electron groups and the geometry associated with it is linear geometry.
(c)
Answer to Problem 5.49EP
The molecular geometry is angular (bent).
Explanation of Solution
Given triatomic molecule is,
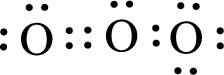
The central atom in the above molecule is found to be oxygen. This has two bonding electron groups and one nonbonding electron groups. The arrangement around the central atom is trigonal planar arrangement. Looking for molecular geometry, the central atom that contains two bonding electron groups and one nonbonding electron groups and it has angular geometry as per VSEPR theory.
Molecular geometry is predicted for the given triatomic molecule using VSEPR theory.
(d)
Interpretation:
The given triatomic molecule geometry has to be predicted using VSEPR theory.
Concept Introduction:
Information about the number of bonds and types of bonds can be obtained from Lewis structure but the molecular geometry cannot be obtained. Three dimensional arrangement of atoms in a molecule can be given by molecular geometry. Physical and chemical properties are determined by the molecular geometry of the molecule.
Using VSEPR theory and Lewis structure, the molecular geometry of the molecule that contain less number of atoms can be predicted. VSEPR theory uses the information from Lewis structure of the molecule to predict the molecular geometry of the molecule. Main concept of VSEPR theory is that electron pairs that are present in the valence shell adopt arrangement in a way that minimize the repulsion between like charges.
If the central atom contains two electron pairs, then it has to be far apart means, it has to be on opposite side of the nucleus. This means the angle has to be
If the central atom contains three electron pairs, then it has to be far apart means, it has to be on corner of a triangle. This means the angle has to be
If the central atom contains four electron pairs, then it has to be far apart means, it has to be in a tetrahedral arrangement. This means the angle has to be
The collection of valence electron that is present in localized region about central atom in a molecule is known as VSEPR electron group. This may contain two electrons, four electrons, or six electrons. The electron group that contain four and six electrons repel each other.
Tetrahedral VSEPR electron group:
The four electron pairs can be of three VSEPR electron groups. They are 4 bonding electron groups, 3 bonding and 1 nonbonding electron groups, and 2 bonding and 2 nonbonding electron groups. The molecular geometry that is associated with 4 bonding electron groups is tetrahedral. The molecular geometry that is associated with 3 bonding and 1 nonbonding electron groups is trigonal pyramidal. The molecular geometry that is associated with 2 bonding and 2 nonbonding electron groups is angular.
Trigonal planar VSEPR electron group:
The three electron pairs can be of two VSEPR electron groups. They are 3 bonding electron groups, and 2 bonding and 1 nonbonding electron groups. The molecular geometry that is associated with 3 bonding electron groups is trigonal planar. The molecular geometry that is associated with 2 bonding and 1 nonbonding electron groups is angular.
Linear VSEPR electron group:
The two electron pairs can be of only one VSEPR electron groups. It is only 2 bonding electron groups and the geometry associated with it is linear geometry.
(d)
Answer to Problem 5.49EP
The molecular geometry is linear.
Explanation of Solution
Given triatomic molecule is,
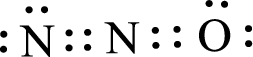
The central atom in the above molecule is found to be nitrogen. This has two bonding electron groups and zero nonbonding electron groups. The arrangement around the central atom is linear arrangement. Looking for molecular geometry, the central atom that contains two bonding electron groups and zero nonbonding electron groups and it has linear geometry as per VSEPR theory.
Molecular geometry is predicted for the given triatomic molecule using VSEPR theory.
Want to see more full solutions like this?
Chapter 5 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Discuss the process that helicases catalyze as well as some of the main components of their mechanics.arrow_forwardA spherical cell with the diameter of 10uMhas a protein concentration of 20 mg/ml. Determine the number of protein molecules within the cell if the molecular weight of an average protein is 50,000 daltons (g/mol). Recall that Avogadro's number is NA 6.0221367×1023 molecules/mol. =arrow_forwardRemember that DNA is a sequence of the letters A,C,T, and G. A binds with T and C binds with G. Solutions are specified by the DNA sequence, such as ACAC or TG, and by the concentration in units of micromolar (uM = 10-M) Consider mixing 30ml of 0.30µM ACAC with 45ml of 0.50pM TG. Predict what reactants and products should be present in the final solution and then check your answer in the virtual lab. (Please give your final answer in total nanomoles (10 moles) for each species in the solution)arrow_forward
- Glucose is the major energy-yielding nutrient for most cells. Assuming a cellular concentration of 1 M (that is, 1 millimole/L), calculate how many molecules of glucose would be present in our hypothetical (and spherical) eukaryotic cell. (Avogadro’s number, the number of molecules in 1 mol of a nonionized substance, is 6.02 × 10 23 .)arrow_forwardTwo common methods of denaturing proteins in the lab are to increase the temperature and/or add chemical denaturants like the detergent SDS (shown below). Describe how each of these processes (heating and addition of SDS) disrupts the forces and interactions that stabilize the native state, leading to unfolding. The answer should include a discussion of the Gibbs free energy of folding, enthalpy, and entropy.arrow_forwardConsider the tripeptide Glu-Asp-Phe. What is the approximate isoelectric of this molecule? In which direction will the tripeptide move in an electric field at pH 1, 5, 10, and 12.arrow_forward
- AH = - 1.2 kcal/mol; Кр 3 16 nM OH 1 HO AH = - 6.0 kcal/mol; Kp = 76 nM OH HOll AH = - 5.5 kcal/mol; Kp = 0.5 nM OH 3 (iv) Consider the molecule 1 being derivatized to yield molecule 2. How would you expect AG for the binding process (to their target) of 2 to compare to 1? Justify your answer.arrow_forwardElectrophoresis is the movement of molecules within a matrix (paper, gel) to which an electric field is applied – from a central origin, a molecule with a net positive charge moves toward the negative pole (often called cathode) and a molecule with a net negative charge moves toward the positive pole (often called anode). for the pentapeptide: N – Lys - gly - asp - his - glu - C What is the net charge at pH 3? 2.What is the net charge at pH 11? 3. At pH 3 will the pentapeptide move to the negative pole, positive pole or not move? 4. At pH 11, the same question.arrow_forwarda Mixtureof the following amino acids is subjected to paper electrophoresis: Gly, Arg and Asp.Which of these amino acids will at (a) pH 2.0 and (b) pH 10 move to the Anode (+) and which will move to the cathode(-)? Motivate your answer.arrow_forward
- According to the configuration of the isostatic, syndiotactic and atactic polymers. Define each of the three configurations and give an example of each of the configurations.(Add the internet link or the bibliographic source where they found the information of the examples)arrow_forwardConsider the tripeptide phenylalanylcysteyl methionine. Part: 0 / 2 Part 1 of 2 Draw the structure of this tripeptide at physiological pH. Click and drag to start drawing a structure. : ☐arrow_forwardConsider the following molecules. (a) Name the molecule and its function for letters: A, C, and E. (b) Name the group and where it is made in the cell for letters: B, C, and D. но H O H O H-N-C-C-N-C-C-N-C-C-OH H CH2 H OH H SH HHHHHHHHHHHHHH H H-C-0 பெ HHHH HHH H HHHHH HHH HHHH -C-C-C-C-C-C-C-C (-C-C-C-C-H H-C-0 OH H 130 HHHHHH HHHH HH HH H C-C-C-C-C-C-(-C-C-C-(-C-C-C-"-H H-C-0 D H. C E.arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
