
Assign a correct set of four quantum numbers for
- (a) Each electron in a nitrogen atom.
- (b) The valence electron in a sodium atom.
- (c) A 3d electron in a nickel atom.
(a)
Interpretation:
A correct set of four quantum numbers for each electron in nitrogen atom has to be given.
Concept Introduction:
Principal Quantum Number (n): In an atom, the electron energy mainly depends on principal quantum number. The energy of an electron becomes lower when the value of n is smaller. The orbital size also depends on n. The size of orbital increases with increase in value of principal quantum number (n)
Angular Momentum Quantum Number (l): It helps to differentiate different shapes of orbitals for given n. For a given n, there are n different shapes of orbitals are present and are denoted as l. Angular momentum quantum number is also known as Azimuthal quantum number. The possible values of angular momentum quantum number are between
Magnetic Quantum Number (
Spin Quantum Number (
Explanation of Solution
The electronic configuration of nitrogen atom is given by
The arrangement of electrons in nitrogen atom is
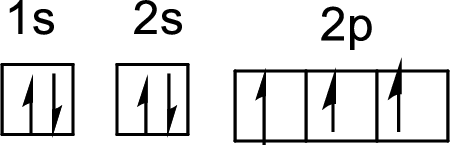
First electron in 1s orbital:
Second electron in 2s orbital:
First electron in 2s orbital:
Second electron in 2s orbital:
First electron in 2p orbital:
Second electron in 2p orbital:
Third electron in 2p orbital:
(b)
Interpretation:
A correct set of four quantum numbers for valence
Concept Introduction:
Refer to part (a)
Explanation of Solution
The electronic configuration of sodium atom is given by
The arrangement of valence

First electron in
(c)
Interpretation:
A correct set of four quantum numbers for a
Concept Introduction:
Refer to part (a)
Explanation of Solution
The electronic configuration of nickel atom is given by
The arrangement of a
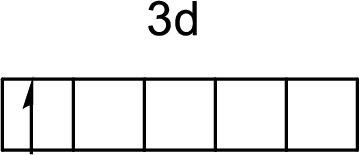
First electron in
Want to see more full solutions like this?
Chapter 5 Solutions
OWLV2 FOR MOORE/STANITSKI'S CHEMISTRY:
- • identify an orbital (as 1s, 3p, etc.) from its quantum numbers, or vice versa.arrow_forwardSuppose that the spin quantum number did not exist, and therefore only one electron could occupy each orbital of a many-electron atom. Give the atomic numbers of the first three noble-gas atoms in this case.arrow_forwardUsing Fig. 2-30, list the elements (ignore the lanthanides and actinides) that have ground-state electron configurations that differ from those we would expect from their positions in the periodic table.arrow_forward
- What experimental evidence supports the quantum theory of light? Explain the wave-particle duality of all matter .. For what size particles must one consider both the wave and the particle properties?arrow_forwardWhat is the difference between an atom’s ground state and an excited state?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





