
(a)
Interpretation:
The period in which sulphur is located in periodic table has to be located.
Concept Introduction:
Period: Horizontal rows in periodic table are called periods. In periodic table, seven periods are present, with each one beginning at the far left.
(a)
Explanation of Solution
A modern periodic table is given by
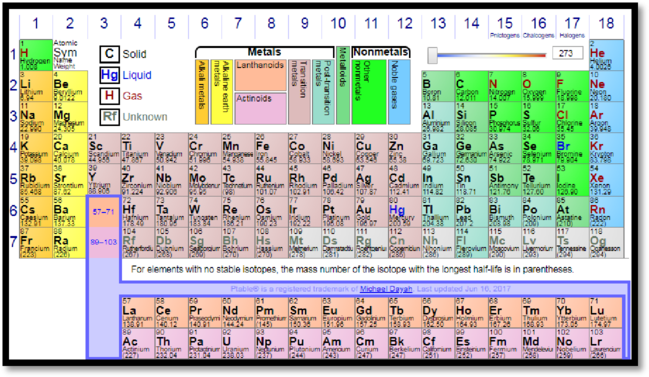
Figure 1
Sulphur belongs to p-block element of periodic table. The outermost electrons present in third shell of the atom. Therefore, sulphur belongs to third period.
(b)
Interpretation:
The complete electron configuration of sulphur has to be written.
Concept Introduction:
Electron configuration: The complete description of the atomic orbitals occupied by all the electrons in an atom or monoatomic ion is known as its electron configuration.
(b)
Explanation of Solution
Sulphur belongs to p-block element of periodic table. It has
(c)
Interpretation:
The atom of an element involved in given reaction has smallest first ionization energy and smallest radius has to be identified.
Concept Introduction:
Electron configuration: The complete description of the atomic orbitals occupied by all the electrons in an atom or monoatomic ion is known as its electron configuration.
Atomic radius: It is defined as distance between nucleus of atom and electron present in outermost valence shell of the atom.
(c)
Explanation of Solution
Given,
The smallest first ionization energy of the three atoms is S, and the smallest radius is O atom. According to periodic trends, elements present in far right of periodic table have highest ionization energy and smallest radius.
(d)
Interpretation:
The mass of chlorine needed to form
(d)
Explanation of Solution
Given,
The mass of chlorine needed is
(e)
Interpretation:
The theoretical mass of thionyl chloride has to be calculated.
(e)
Explanation of Solution
Given,
The mass of thionyl chloride formed is
Want to see more full solutions like this?
Chapter 5 Solutions
OWLV2 FOR MOORE/STANITSKI'S CHEMISTRY:
- (1) What is the valence electron configuration for the arsenic atom? (2) What is the valence electron configuration for the fluorine atom?arrow_forward(a) Identify the number of electrons in the ground-state outer shell of atomic oxygen (atomic number 8).(b) How many electrons are in the ground-state outer shell of fluorine?arrow_forwardRubidium and bromine atoms are depicted at right. (a) What monatomic ions do they form? (b) What electronic feature characterizes this pair of ions, and which noble gas are they related to? (c) Which pair best represents the relative ionic sizes?arrow_forward
- By gaining 2 electrons, a sulfur atom attains an electron arrangement like that of the noble gas argon. (a) Explain why the sulfur atom does not become an argon atom. (b) State what the sulfur atom does become when it gains 2 electrons.arrow_forwardBoron, atomic number 5, occurs naturally as two isotopes, 10B and 11B, with natural abundances of 19.9% and 80.1%, respectively.(a) In what ways do the two isotopes differ from each other? Does the electronic configuration of 10B differ from that of 11B? (b) Drawthe orbital diagram for an atom of 11B. Which electrons are the valence electrons? (c) Indicate three ways in which the 1s electrons inboron differ from its 2s electrons. (d) Elemental boron reacts with fluorine to form BF3, a gas. Write a balanced chemical equation forthe reaction of solid boron with fluorine gas. (e) ΔHf° for BF31g2 is -1135.6 kJ>mol. Calculate the standard enthalpy change in thereaction of boron with fluorine. (f) Will the mass percentage of F be the same in 10BF3 and 11BF3? If not, why is that the case?arrow_forward(1) What is the valence electron configuration for the neon atom? (2) What is the valence electron configuration for the sulfur atom?arrow_forward
- Give the shell electron configuration for the following. (For example, the shell electron configuration of lithium is written 2,1.) (a) argon (b) magnesiumarrow_forward7. The first ionisation energy of the chlorine atom is 1251 kJ/mol. Without looking at your text book, say which of the following values would be the more likely ionisation energy for the iodine atom. Explain your choice. (a) 1000 kJ/mol (b) 1400 kJ/molarrow_forward15. (a) b) Identify the element that is described by the following information. Refer to a periodic table if necessary. It is a group 14 (III A) metalloid in the 3rd period. It is a group 15 (VA) metalloid in the 5th period. It is the other metalloid in group 15 (VA). d) It is a halogen that exists in the liquid state at room temperature. 16. What is the relationship between electron arrangement and the organization of elements in the periodic table?arrow_forward
- 1.) Which element would have the lowest ionization energy? S, Ge, Al, Ca, Rb 2.) Which equation below illustrates the second ionization energy for sodium A) Na (g) + 2e– → Na–2 (g) B) Na (g) → Na+2 (g) + 2e– C) Na+ (g) →Na+2(g) + e– D) Na+2 (g) + 2e– → Na (g) E) Na (g) → Na+ (g) + e– F) Na (g) + e– → Na– (g)arrow_forwardHow many valence electrons are in each element?(a) Ba (b) Al (c) Be (d) Searrow_forward(a) Why do successive IEs of a given element always increase? (b) When the difference between successive IEs of a given element is exceptionally large (for example, between IE1 and IE2 of K), what do we learn about its electron configuration? (c) The bars represent the relative magnitudes of the first five ionization energies of an atom: Identify the element and write its complete electron config uration, assuming it comes from (a) Period 2; (b) Period 3; (c) Period 4.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

