
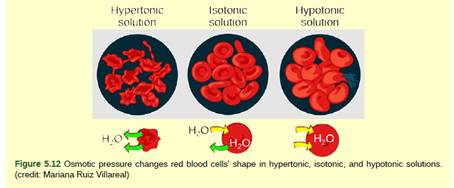
Figure 5.12 A doctor injects a patient with what the doctor thinks is an isotonic saline solution. The patient dies, and an autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?
To analyze:
The type of the solution inserted in the body of the patient, which causes the destruction of the RBCs.
Introduction:
Solutions are of three types depending upon the concentration of solutes. The solution having a high concentration of solutes is called hypertonic solution, the one with less solute is called hypotonic solution and two solutions having equal concentration of solutes is known as an isotonic solution.
Explanation of Solution
The solution which is injected into a patient was not isotonic but hypotonic. A hypotonic solution is the one which has less solute and more solvent (water) concentration. Due to this, the solvent rushes inside the RBC by the process called as diffusion. When the water rushes in the RBCs, the cells swell and burst.
Diffusion of solutes and water takes place from high concentration to low concentration. The RBCs, when placed in the hypotonic solution swell and burst, which causes the destruction of RBCs.
Want to see more full solutions like this?
Chapter 5 Solutions
Biology 2e
Additional Science Textbook Solutions
College Physics
Campbell Biology in Focus (2nd Edition)
Human Anatomy & Physiology
Campbell Essential Biology with Physiology (5th Edition)
Concepts of Genetics (12th Edition)
Campbell Essential Biology (6th Edition) - standalone book
- Figure 3.22 A doctor injects a patient with what he thinks is isotonic saline solution. The patient dies, and autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?arrow_forwardWhy is it advantageous for the cell membrane to be fluid in nature?arrow_forwardFigure 4.8 If the nucleolus were not able to carry out its function, what other cellular organelles would be affected?arrow_forward
- Does consuming too much water create hypertonic, hypotonic or isotonic environment in the body fluid surround your cells? How would this affect the cells?arrow_forwardEach diagram shows a cell with a semipermeable membrane submerged in a solution. Solutions contain different amounts of solutes (glucose, sodium). The membrane is NOT permeable to glucose or sodium.arrow_forwardA human cell was placed in a solution. The human cell absorbed the water and then burst. What kind of solution was the human cell placed in?arrow_forward
- Elastic nature of plasma membrane is due to the presence of (a) lipids (b) proteins (c) carbohydrates (d) waterarrow_forwardOsmosis is a special type of diffusion that ________. a. moves water from an area with a high solute concentration to one of lower solute concentration b. moves sugar from an area with a high solute concentration to a lower solute concentration c. moves water from an area with a low solute concentration to one of higher solute concentrationarrow_forwardThe movement of molecules that expends energy to move potassium ions into the cell and a different number of sodium ions out of the cell Simple Diffusion Facilitated Diffusion Osmosis Sodium-Potassium Pumparrow_forward
- Osmosis is _________________ movement of water down its concentration gradient using a protein channel movement of materials against their concentration gradient movement of one substance against, and one substance down their concentration gradients movement of molecules from high concentration to low concentration using no protein channels movement of materials down their concentration gradient using a protein channelarrow_forwardOsmosis is water movement across a semipermeable membrane. Which of the following is true about water movement across cell membranes? A. In a hypotonic solution, cells will swell. B. In an isotonic solution, cells will shrink. C. In a hypertonic solution, cells will stay the same. D. Cells can neither shrink nor swell because water cannot penetrate the plasma membrane.arrow_forwardHow many cells are in a human bodyarrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning




