
The ingenious Stirling engine is a true
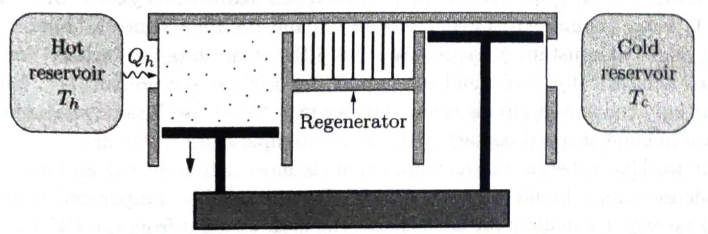
Figure 4.7. A Stirling engine, shown during the power stroke when the hot piston is moving outward and the cold piston is at rest. (For simplicity, the linkages between the two pistons are not shown.)
Trending nowThis is a popular solution!

Chapter 4 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Physics (5th Edition)
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Applied Physics (11th Edition)
Life in the Universe (4th Edition)
Physics for Scientists and Engineers with Modern Physics
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
- A rubberized sphere contains carbon dioxide. If the initial radius of the sphere is 15cm: How much work is done if the carbon dioxide-filled sphere expanded to five times its original volume against the pressure at STP? Express your answer in L atm. b. Upon the expansion of the sphere, what is the kinetic energy of a single carbon dioxide molecule if it travels across the diameter and covers it in 30ms? Assume that the sphere contains one mole of carbon dioxide. Express your answer in J. How much power can all the molecules in this sphere generate after 30s? Assume that the sphere contains one mole of carbon dioxide and that all molecules have the same kinetic energy. Express your answer in W. (Use the KE obtained from the previous required) 2. A rubberized, cubic chamber was filled with butane gas and was expanded from 3.0L to 4.5L at a pressure of 2.7atm and it absorbed 230J of heat from the surroundings. a. What is the change in the energy of the system? Express your answer in L…arrow_forwardPlease describe to me the laws of thermodynamics. There are a certain number of them and therefore you must have all of them. Each should be labeled with it's number. For instance "The First Law of thermodynamics says that..."arrow_forwardConverting sunlight to electricity with solar cells has an efficiency of 15%. It's possible to achieve a higher efficiency (though currently at higher cost) by using concentrated sunlight as the hot reservoir of a heat engine. Each dish in (Figure 1) concentrates sunlight on one side of a heat engine, producing a hot-reservoir temperature of 560 ∘C. The cold reservoir, ambient air, is approximately 30 ∘C. The actual working efficiency of this device is 30%. What is the theoretical maximum efficiency?arrow_forward
- Imagine that you are rolling three typical six-sided dice. Each way that you can roll a particular outcome using these three dice represents a microstate for that outcome. How many ways can you roll a five with these three dice? That is, how many microstates exist for a roll of five with three dice? What is the entropy associated with an outcome of five in this situation?arrow_forwardA thermodynamic system is taken from state A to state B to state C, and then back to A, as shown in the p-V diagram of Figure below.The vertical scale is set by p, = 40 Pa, and the horizontal scale is set by Vs = 4.0m³. i. Analyze the graph using first law of thermodynamics and complete the following table by inserting a plus sign, minus sign, or a zero in each indicated cell ? AEint A >B (a) (b) + (c) (d) + (e) (f) (3) V, Volume (m) ii. Analyze the graph using first law of thermodynamics and determine the net work done by the system as it moves once through the cycle ABCA?arrow_forwardLet’s say that the Carnot engine takes 100 J of heat from a reservoir at 500 K, does some work, and discards some heat at a reservoir at 300 K. How much work does it do, how much heat is discarded, and what is its efficiency?arrow_forward
- 0.5 moles of cinnamaldehyde gas that has an initial volume of ten liters expands under the following conditions: 185°F and 800mmHg external pressure. a. How much work is done? Express your answer in L atm. b. Assuming that the container is cylindrical with a base radius of 10cm and can only expand vertically, how much kinetic energy does a single molecule possesses if it travels from base to base of the expanded cylinder in 10us? Express your answer in Jarrow_forward‼️Illustrate the problem ONLY: A rubberized sphere contains carbon dioxide. If the initial radius of the sphere is 15cm: a. How much work is done if the carbon dioxide-filled sphere expanded to five times its original volume against the pressure at STP? Express your answer in L atm. b. Upon the expansion of the sphere, what is the kinetic energy of a single carbon dioxide molecule if it travels across the diameter and covers it in 30ms? Assume that the sphere contains one mole of carbon dioxide. Express your answer in J. b. How much power can all the molecules in this sphere generate after 30s? Assume that the sphere contains one mole of carbon dioxide and that all molecules have the same kinetic energy. Express your answer in W. (Use the KE obtained from the previous required)‼️Illustrate the problem ONLYarrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 422 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy? Jarrow_forward
- The temperature in the deep interiors of some giant molecular clouds in the Milky Way galaxy is 50 K. Compare the amount of energy that would have to be transferred to this environment to the amount that would have to be transferred to a room temperature environment to bring about a 7.7 J/K increase in the entropy of the universe in each case. ΔEroom temp/ ΔEMilky Way =arrow_forwardIn (Figure 1), consider the closed loop 1→2→3→4→1. This is a cyclic process in which the initial and final states are the same. Use values p0 = 0.10 atm and V0 = 0.50 L . Part A: Find the total work done by the system in this process, and show that it is equal to the area enclosed by the loop. (answer in J) Part B: How is the work done during the process in Part A related to the work done if the loop is traversed in the opposite direction, 1→4→3→2→1?arrow_forwardA hand-driven tire pump has a piston with a 2.00 cm diameter and a maximum stroke of 36.0 cm. (a) How much work (in J) do you do in one stroke if the average gauge pressure is 2.40 x 10° N/m (about 35 psi)? J (b) What average force (in N) do you exert on the piston, neglecting friction and gravity? N Additional Materials Reading ES Scanned with CamScanner Submit Answerarrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





