
Concept explainers
Although styrene undergoes both cationic and anionic
a. 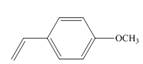 c.
c. 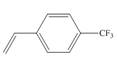
b. 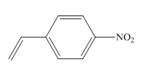 d.
d. 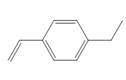
(a)
Interpretation: The method, anionic or cationic polymerization preferred with the given substituted styrene is to be identified, and an explanation corresponding to it is to be stated.
Concept introduction: An alkene
Answer to Problem 30.43P
The given substituted styrene prefers to undergo cationic polymerization, due to presence of electron releasing group on benzene ring.
Explanation of Solution
The given substituted styrene resembles to
An alkene
An electron releasing group releases electron density towards benzene ring and stabilizes carbocation formed during the cationic polymerization, whereas an electron withdrawing group withdraws electron density towards itself and stabilizes carbanion formed during the anionic polymerization.
Since the given substituted styrene involves bonding of an electron releasing group to benzene ring, it prefers to undergo cationic polymerization.
(b)
Interpretation:The method, anionic or cationic polymerization preferred with the given substituted styrene is to be identified, and an explanation corresponding to it is to be stated.
Concept introduction: An alkene
Answer to Problem 30.43P
The given substituted styrene prefers to undergo anionic polymerization, due to presence of electron withdrawing group on benzene ring.
Explanation of Solution
The given substituted styrene resembles to
An alkene
An electron releasing group releases electron density towards benzene ring and stabilizes carbocation formed during the cationic polymerization, whereas an electron withdrawing group withdraws electron density towards itself and stabilizes carbanion formed during the anionic polymerization.
Since the given substituted styrene involves bonding of an electron withdrawing group to benzene ring, it prefers to undergo anionic polymerization.
(c)
Interpretation:The method, anionic or cationic polymerization preferred with the given substituted styrene is to be identified, and an explanation corresponding to it is to be stated.
Concept introduction: An alkene
Answer to Problem 30.43P
The given substituted styrene prefers to undergo anionic polymerization, due to presence of electron withdrawing group on benzene ring.
Explanation of Solution
The given substituted styrene resembles to
An alkene
An electron releasing group releases electron density towards benzene ring and stabilizes carbocation formed during the cationic polymerization, whereas an electron withdrawing group withdraws electron density towards itself and stabilizes carbanion formed during the anionic polymerization.
Since the given substituted styrene involves bonding of an electron withdrawing group to benzene ring, it prefers to undergo anionic polymerization.
(d)
Interpretation:The method, anionic or cationic polymerization preferred with the given substituted styrene is to be identified, and an explanation corresponding to it is to be stated.
Concept introduction: An alkene
Answer to Problem 30.43P
The given substituted styrene prefers to undergo cationic polymerization, due to presence of electron releasing group on benzene ring.
Explanation of Solution
The given substituted styrene resembles to
An alkene
An electron releasing group releases electron density towards benzene ring and stabilizes carbocation formed during the cationic polymerization, whereas an electron withdrawing group withdraws electron density towards itself and stabilizes carbanion formed during the anionic polymerization.
Since the given substituted styrene involves bonding of an electron releasing group to benzene ring, it prefers to undergo cationic polymerization.
(a) The given substituted styrene prefers to undergo cationic polymerization, due to presence of an electron releasing group on benzene ring.
(b) The given substituted styrene prefers to undergo anionic polymerization, due to presence of an electron withdrawing group on benzene ring.
(c) The given substituted styrene prefers to undergo anionic polymerization, due to presence of an electron withdrawing group on benzene ring.
(d) The given substituted styrene prefers to undergo cationic polymerization, due to presence of an electron releasing group on benzene ring.
Want to see more full solutions like this?
Chapter 30 Solutions
Organic Chemistry
- 1 please answer the question correctly. draw the polymer and name the polymerisation process for each case. will give a thumbs up if correct. thank you.arrow_forwardDraw the structure of the alternating copolymer formed from each pair of monomers. a. CN and and с. CN and b. and d. andarrow_forward2) For the monomer given determine what type of polymerization (radical, anionic, cationic, and/or step-wise) it will undergo most readily or if it will not undergo polymerization at all. Explain your answer. A.arrow_forward
- Which of the following molecules can be used as a monomer to generate this polymer? So the answer's B... im having quite the trouble understanding where in the polymer the double bond broke. Could someone explain and draw in EXTREME details how the answer's B and not any other answer choice?arrow_forward(d) show termination and propogation process in polyethylene terephthalate (PET).arrow_forward1.- How does the molecular weight change vs % conversion in a step polymerization? Explain this behavior and why does it occur? 2.- Name at least 3 requirements for obtaining a high molecular weight polymer by step polymerization. 3.- How can you minimize cyclization reactions in a step polymerization? Explain with equations and narrative Why do you want to minimize cyclization? Is it possible to eliminate cyclic products completely?arrow_forward
- Write equations for the following reactions: a. Linear polymerization of formaldehyde b. Cyclic polymerization of formaldehyde c. Linear polymerization of acetaldehyde d. Cyclic polymerization of acetaldehydearrow_forwardConsider monomers A–C. (a) Rank the monomers in order of increasingreactivity in cationic polymerization. (b) Rank the monomers in order ofincreasing reactivity in anionic polymerization.arrow_forwardIn the sentence below, fill in the blanks with appropriate terms. Poly (ethylene terephthalate) is a .., obtained by polymerization of and yunun - .. .. aratle A polyester, addition, dicarboxylic acids, polyalcohols. B polyamide, condensation, diamines, dicarboxylic acids. polyester, condensation, dicarboxylic acids, polyalcohols. D polyamide, condensation, dicarboxylic acids, polyalcohols.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




