
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 59P
a. How many primary carbons does each of the following compounds have?
b. How many secondary carbons does each one have?
c. How many tertiary carbons does each one have?
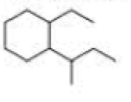
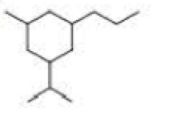
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider 1,2-dimethylcyclohexane.a. Draw structures for the cis and trans isomers using a hexagon for the six-membered ring.b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable?c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable?d. Which isomer, cis or trans, is more stable and why?
How many secondary carbons does each one have?
How are the molecules below related to each other? (Hint: convert each to a
partially condensed structual formula).
Н.
H
H
CH3
H₂C
CH3
CH3
and
H
H
H
H₂c-CH₂
CH3
H
CH3
Select one:
OA. They are constitutional isomers.
OB. They are identical compounds.
O C. They are geometrical isomers.
O D. They are stereoisomers.
O E. They are different compounds, not isomers..
Chapter 3 Solutions
Organic Chemistry (8th Edition)
Ch. 3.1 - Name each of the following:Ch. 3.1 - Draw the structure of a compound with molecular...Ch. 3.1 - Draw the structures and name the four...Ch. 3.1 - Prob. 6PCh. 3.1 - Draw the structure for each of the following: a....Ch. 3.1 - Name the following compounds: a. CH3OCH2CH3 b....Ch. 3.2 - Prob. 9PCh. 3.2 - Draw the structure for each of the following: a....Ch. 3.2 - Give each substituent on the nine-carbon chain a...Ch. 3.2 - Prob. 14P
Ch. 3.3 - What is each compounds systematic name?Ch. 3.3 - Prob. 16PCh. 3.3 - Prob. 17PCh. 3.3 - Prob. 18PCh. 3.3 - Prob. 19PCh. 3.4 - Give two names for each of the following alkyl...Ch. 3.4 - Prob. 21PCh. 3.5 - a. What is each ethers systematic name? 1....Ch. 3.6 - Give each of the following a systematic name and...Ch. 3.6 - Draw the structures of a homologous series of...Ch. 3.6 - Prob. 25PCh. 3.6 - Prob. 26PCh. 3.7 - Prob. 27PCh. 3.7 - Are the following compounds primary, secondary, or...Ch. 3.7 - Draw condensed and skeletal structures for each of...Ch. 3.7 - For each of the following, give the systematic...Ch. 3.8 - Predict the approximate size of the following bond...Ch. 3.9 - Prob. 32PCh. 3.9 - Prob. 33PCh. 3.9 - Prob. 34PCh. 3.9 - Rank the following compounds from highest boiling...Ch. 3.9 - Rank the compounds in each set from highest...Ch. 3.10 - In which solvent would cyclohexane have the lowest...Ch. 3.10 - Prob. 38PCh. 3.10 - Prob. 39PCh. 3.11 - a. Draw all the staggered and eclipsed conformers...Ch. 3.11 - Prob. 41PCh. 3.11 - Using Newman projections, draw the most stable...Ch. 3.12 - The bond angles in a regular polygon with n sides...Ch. 3.12 - Prob. 44PCh. 3.13 - Draw 1,2,3,4,5,6-hexachlorocydohexane with a. all...Ch. 3.14 - Using the data in Table 3.9, calculate the...Ch. 3.14 - The chair conformer of fluorocyclohexane is 0.25...Ch. 3.15 - Prob. 48PCh. 3.15 - Which has a higher percentage of the...Ch. 3.15 - a. Draw the more stable chair conformer of...Ch. 3.15 - For each of the following disubstituted...Ch. 3.15 - a. Draw Newman projections of the two conformers...Ch. 3.15 - a. Calculate the energy difference between the two...Ch. 3 - a. How many hydrogen does an alkene with 17...Ch. 3 - Draw the structure of octane and isooctaneCh. 3 - Draw a condensed structure and a skeletal...Ch. 3 - Prob. 56PCh. 3 - a. What is each compounds systematic name? b. Draw...Ch. 3 - Which of the following represents a cis isomer?Ch. 3 - a. How many primary carbons does each of the...Ch. 3 - Which of the following conformers of isobutyl...Ch. 3 - Draw a skeletal structure for an alkane that has...Ch. 3 - What is each compounds systematic name? a....Ch. 3 - Which bus a. the higher boiling point:...Ch. 3 - a. Draw Newman projections of the two conformers...Ch. 3 - Ansaid and Motrin belong to the group of drugs...Ch. 3 - Prob. 66PCh. 3 - A student was given the structural formulas of...Ch. 3 - Which of the following conformers has the highest...Ch. 3 - Prob. 69PCh. 3 - Draw skeletal structures for the following: a....Ch. 3 - For rotation about the C-3C-4 bond of...Ch. 3 - Prob. 72PCh. 3 - What is each compounds systematic name? a. b. c....Ch. 3 - Draw the two chair conformers for each of the...Ch. 3 - Why are lower molecular weight alcohols more...Ch. 3 - a. Draw a potential energy diagram for rotation...Ch. 3 - For each of the following compound, determine...Ch. 3 - How many ethers have molecular formula C5H12O?...Ch. 3 - Draw the most stable conformer of the following...Ch. 3 - What is each compounds systematic name?Ch. 3 - Calculate the energy difference between the two...Ch. 3 - The most stable from of glucose (blood sugar) is a...Ch. 3 - What is each compound s systematic name?Ch. 3 - Explain the following: a. 1-Hexanol has a higher...Ch. 3 - One of the chair conformers of cis-...Ch. 3 - Bromine is a larger atom than chlorine, but the...Ch. 3 - Name the following compounds:Ch. 3 - Prob. 88PCh. 3 - Using the data obtained in Problem 85, calculate...Ch. 3 - Draw the conformers for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Name each compound and determine the relationship between the pairs of compounds. O a. Name: b. Name: N C. Relationship between structures above: Conformational isomer or Structural isomer 2. Name: 3. Is the following structure the cis stereoisomer or the trans stereoisomer?arrow_forward9) There are 3 different cyclopropane molecules with the formula GHĄC12. a. Draw and build the 3 molecules. b. Below each drawing, name each molecule with correct nomenclature. Label a pair that are constitutional isomers. d. Label a pair that are stereoisomers (or configurational isomers). С.arrow_forwardThe given compound, 2-methylbutane is having A. One primary, one secondary and three tertiary carbons B. Three secondary, one primary and one tertiary carbons C. Three primary, one secondary and one tertiary carbons D. One primary, three secondary and one tertiary carbonsarrow_forward
- Part A What is the relationship between the following molecules? CH,CH3 CH3 CH2 CHCH3 CH;CH2-CH- CH-CH,-CH-CH-CH3 CH3 CH,CH3 CH3 CHCH3 CH,CH3 CH;CH, CH-CH-CH,-CH–ĊH-CH,CH,CH3 CH3 CH3 O They are different molecules which are not isomers. O They are identical. O They are isomers of each other. O none of the above Submit Request Answerarrow_forward13. How many tertiary carbons and secondary hydrogens does the compound shown below contain? A. 5 tertiary carbons and 10 secondary hydrogens. B. 5 tertiary carbons and 5 secondary hydrogens. C. 3 tertiary carbons and 10 secondary hydrogens. D. 3 tertiary carbons and 12 secondary hydrogens. oulaarrow_forwardThe dimethylcyclohexane with the structure shown below is: ÇH3 CH3 Select one: O a. a trans isomer with the CH3 groups in axial positions. O b. a cis isomer with the CH3 groups in equatorial positions. O c. a cis isomer with the CH3 groups in equatorial and axial positions. O d. a trans isomer with the CH3 groups in equatorial positions.arrow_forward
- How many hydrogens do the following compound have? C9H?NO, has one ring and three double bond a. 13 b. 16 c. 12 d. 11arrow_forwarda. Draw the lowest energy conformation for the compound shown in the image. b. Draw the complete structural formula of the following compound: cis-1-bromo-3-chlorocyclohexane.arrow_forwardtof How are the molecules below related to each other? (Hint: convert each to a partially condensed structual formula). CH3 Н. HY H₂C CH3 CH3 and Н. H Select one: O A. They are stereoisomers. H H HỌC CHI CHO CH3 OB. They are constitutional isomers. d They are identical compounds. They are different compounds, not isomers.. OD. O E. They are geometrical isomers. Why does acetone [(CH3)2C=0, dipole moment = 2.69 D] have a larger dipole moment than phosgene [Cl₂C=0, dipole moment 1.17 D]? Note: electronegativities C = 2.5, Cl = 3.2, 0 = 3.5, H = 2.2arrow_forward
- 6. Consider the structure of cis-1,2-dimethylcyclopropane and trans-1,2-dimethylcyclopropane A. Which compound is more stable? Explain. B. Which compound will release more heat upon combustion? Explain C. Based on the following interactions, predict the difference in energy between the cis and trans isomers. Each CH3 Each CH3 Each H Each H CH3 gauche interaction is worth 0.9 kcal/mol CH3 eclipsed interaction is worth 2.6 kcal/mol CH3 eclipsed interaction is worth 1.4 kcal/mol H eclipsed interaction is worth 1.0 kcal/molarrow_forwardConsider 1,2-dimethylcyclohexane. a.Draw structures for the cis and trans isomers using a hexagon for the sixmembered ring. b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable? c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable? d.Which isomer, cis or trans, is more stable and why?arrow_forwardHow many primary, secondary, tertiary, and quarternary carbons are there in the molecule below?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY