
Biological Science (6th Edition)
6th Edition
ISBN: 9780321976499
Author: Scott Freeman, Kim Quillin, Lizabeth Allison, Michael Black, Emily Taylor, Greg Podgorski, Jeff Carmichael
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 15PIAT
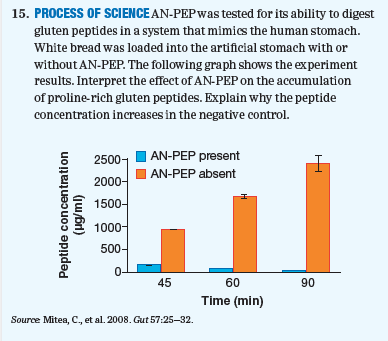
PROCESS OF SCIENCE AN-PEP was tested for its ability to digest gluten peptides in a system that mimics the human stomach. White bread was loaded into the artificial stomach with or without AN-PEP. The following graph shows the experiment results. Interpret the effect of AN-PEP on the accumulation of proline-rich gluten peptides. Explain why the peptide concentration increases in the negative control.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
You are studying the uptake of L-leucine by epithelial cells of the mouse intestine. Measurements of the rates of uptake of L-leucine and severalof its analogs, with and without Na+ in the assay buffer, yield the results given in the table below. What can you conclude about the properties and mechanism of the leucine transporter? Would you expect L-leucine uptake to be inhibited by ouabain?
You have expressed a protein of interest in E. coli cells for further study in the lab. The protein has a net positive charge at pH 6, absorbs UV light at 280m, and has insulin binding activity.Briefly describe a purification scheme with at least three steps that will leverage these properties and generate pure protein.
Small molecules are used as inhibitors of protein action - as drugs. They most often do this
by blocking the active site within the protein. Potential drugs can be screened
computationally to determine if they are strongly bound to the protein.
Figure 1 shows a possible conformation of a candidate drug molecule, 4-bromo-2-
carboxymethylamide-pyrrole (abbreviation: BCMAP) at the active site of a protein
(abbreviation: PR). Figure 2 shows the full protein structure whilst figure 3 shows a known
inhibitor of the protein at the site, overlayed with another calculated conformer of BCMAP.
(a) Explain what types of interactions, both intermolecular and intramolecular, that a
molecular mechanics forcefield must be able to describe in order to be able to
accurately determine the geometry of BCMAP in the protein. Identify which
interactions will be the most important to describe accurately.
Figure 1.4-bromo-2-carboxymethylamide-pyrrole (BCMAP) (C, N, O, and Br atoms in yellow, blue,
red, and…
Chapter 3 Solutions
Biological Science (6th Edition)
Ch. 3 - 1. What two functional groups are bound to the...Ch. 3 - 2. What type of bond is directly involved in the...Ch. 3 - What type of information is used to direct...Ch. 3 - 4. What is an active site?
a. the location in an...Ch. 3 - Prob. 5TYUCh. 3 - Prob. 6TYUCh. 3 - 7. Why are proteins not considered to be a good...Ch. 3 - Prob. 8TYUCh. 3 - Prob. 9TYPSSCh. 3 - 10. Make a concept map (see BioSkills 12) that...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- b. Compounds A, B, C, and D are known to be intermediates in the pathway for production of protein E. To determine where the block in protein-E production occurred in each individual, the various intermediates were given to each individuals cel Is in culture. After a few weeks of growth with the intermediate, the cells were assayed for the production of protein E. The results for each individuals cells are given in the following table. A plus sign means that protein E was produced after the cells were given the intermediate listed at the top of the column. A minus sign means that the cells still could not produce protein E even after being exposed to the intermediate at the top of the column. Denote the point in the pathway in which each individual is blocked.arrow_forwarda. Compounds A, B, C, and D are known to be intermediates in the pathway for production of protein E. To determine where the block in protein-E production occurred in each individual, the various intermediates were given to each individuals cel Is in culture. After a few weeks of growth with the intermediate, the cells were assayed for the production of protein E. The results for each individuals cells are given in the following table. A plus sign means that protein E was produced after the cells were given the intermediate listed at the top of the column. A minus sign means that the cells still could not produce protein E even after being exposed to the intermediate at the top of the column. Draw the pathway leading to the production of protein E.arrow_forwardYou are working with a protein that is known to form an functioning complex of three identical polypeptide subunits, each with a binding site for a ligand of interest. You are studying an inherited mutation in the protein that changes one Asp residue to a Leu residue in each subunit. A comparison of the binding curves for the normal (wild-type) and mutant versions of the complex are shown below. Suggest a mechanism for the effect that this mutation has on the structure of the complex.arrow_forward
- Suppose that you are tasked to determine the protein concentration of an unknown protein solution via Bradford assay. You prepared six solutions of bovine serum albumin (BSA) with different concentrations. The initial concentration of the BSA stock solution is 7.50 mg/mL. Approximately 200 µL of Bradford Reagent was added to each of these solutions and the absorbance at 595 nm was taken after 5 minutes. See the table below for data on the standard solutions. Standard # A595 BSA conc (mg/mL) 0.000 0.158 2 1.125 0.291 2.250 0.372 4 3.375 0.503 5 4.500 6 5.625 0.675 Determine the protein concentration, in mg/mL, of the unknown solution if its absorbance at 595 nm was 0.248. Note: Final answer format must be x.xx (two decimal places). Round off only in the final answer. Do not round off in the middle of calculation.arrow_forwardA tetrapeptide, glutamate-glycine-alanine-lysine, is prepared at at concentration of 1 mM (0.001 M) and is measured in the standard setup (pathlength of 1 cm). What is the approximate absorbance of this peptide at 280 nm? Hint: if the peptide contained a single tryptophan, the answer would be about 10. 10 280 1 0arrow_forwardAn active site of a hypothetical serine protease with a peptide substrate bound is shown below: This serine protease has 3 specificity pockets (S1, S2, S1') as shown in the figure above. S1 pocket has a glutamic acid in the bottom, the S2 pocket is small and hydrophobic, and the S1' pocket is deep and hydrophobic. Based on this information you can conclude that R1 is most likely side chain of a. Asp b. Lys c. Lle d. Gly e. Phearrow_forward
- Consider, for example, that a particular serine residue is phosphorylated to activate the protein. In the lab, a similar effect is achieved by mutating this residue to either a D (aspartic acid) or E (glutamic acid). What could be the reason?arrow_forwardA binding curve for the binding of the amino acid tryptophan to the protein called TxtE is shown below. Estimate the association equilibrium constant (Ka; in units of M-1) for Trp binding to TxtE.arrow_forwardSubstrate A has a Km of 45uM and a kcat of 100/s with trypsin and a Km of 540mM and a kcat of 2/s with chymotrypsin. Substrate A is a better substrate of [trypsin/chymotrypsin] and likely contains a/an/some [aromatic/acidic/basic] residues(s) in its sequence. Chymotrypsin contains a [glycine/serine/aspartate] in its specificity pocket which likely [repel/attract] the residues in substrate A.arrow_forward
- H CH₂ H₂C HC-CH3 CH₂ H H₂C (S) H₂C H CH₂ CH₂ CH₂ NH O C NH NH₂ a) Which of the following statements about this peptide are correct? Group of answer choices Treatment of this peptide with trypsin generates two products. This peptide is a substrate for carboxypeptidase A Treatment of this peptide with cyanogen bromide generates a pentapeptide and a tripeptide. Treatment of this peptide with chymotrypsin generates three products. Treatment of this peptide with elastase generates 2 products. None of the above statements are correct. b) What is the sequence of this peptide using one letter abbreviations? c) What is the pH which would correspond to the ionization of the peptide as drawn above? 1, 5, 7, 10, 14arrow_forwardThe hydropathy plot of a particular hyaluronan synthase is plotted by Heldermon et al and is shown below. From this plot, Heldermon et al circled six putative (or predicated) membrane domains (PMDs). Circle the six regions in the plot above you believe could be transmembrane domains of the protein and indicate why you chose those regions. Please include a description of what information a hydropathy plot gives researchers.arrow_forwardIn the protein denaturation experiment, which of the following can be a consequence of the air bubbles in the viscometry run for an aqueous solution of protein with a denaturant?a. Increased t0b. Decreased t0c. Increased nspd. Decreased nspAll of the statements about protein denaturation are true EXCEPT:a. The viscosity of linear proteins is greater than that of the globular proteins.b. Only the BME can disrupt a covalent bond while the other denaturants can just disrupt non-covalent bonds.c. Protein renaturation is possible but in some cases protein denaturation proceeds to protein degradation. d. Protein denaturation generally disrupts tertiary or quaternary structures onlyarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning

Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning
Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License