
Concept explainers
Interpretation:
[1,3]sigmatropic migrations of hydrogen cannot occur under thermal conditions but [1,3] sigmatropic migration of carbon can. Reason for this should be explained.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
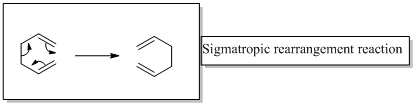
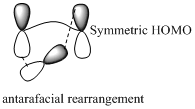
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3. Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction. When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state. Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital.
Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting

Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- (a): Calculate Miller's indices in the hexagonal structure of its intersections. aj = 1, a2= -1/2, a3 = 1 , c= ∞ and draw it.arrow_forwardHow many normal modes (different types of vibrational motion) are predicted for the following substances (a) Ar (b) CO (c) CO2 (d) NH3?arrow_forwardDescribe the formation of a hydrogen bond in terms of (a) electrostatic interactions and (b) molecular orbitals. How would you identify the better model?arrow_forward
- Calculate f(E), the probability predicted by the Fermi–Dirac distribution, for the case where the energy E is the thermal energy, kT, above the Fermi energy: E = EF + kT.arrow_forwardProton 1 moves with a speed v from theeast coast to the west coast in the continental United States; proton 2 moves with the same speed from the southern United Statestoward Canada. (a) Is the magnitude of the magnetic force experienced by proton 2 greater than, less than, or equal to the forceexperienced by proton 1? (b) Choose the best explanation fromamong the following:I. The protons experience the same force because the magneticfield is the same and their speeds are the same.II. Proton 1 experiences the greater force because it moves atright angles to the magnetic field.III. Proton 2 experiences the greater force because it moves in thesame direction as the magnetic field.arrow_forwardAmong TiCl4, VCl3 and FeCl2- which one will be drawn more strongly into a magnetic field and why?arrow_forward
- 9. Consider the ethyl iodide molecule, CH CH₂, which of the statements below is correct? (A) CH, protons are more deshielded. (B) The energy gap between a and ß spin states of CH₂ protons is smaller than the counterpart of CH; protons. (C) The electronegative element I shields the CH₂ protons. (D) None of the above.arrow_forward2. Molecules absorb IR radiation consistent with vibrational energy and rotational energy. Which of these is present in condensed phases (liquid, solution, solid)?arrow_forwardEstimate the dipole moment of (a) an HF molecule. (b) an HCI molecule. and (c) an HBr molecule from the electronegativities of the elements. Express your answers in debye and coulomb-metres.arrow_forward
- We can use the classical harmonic oscillator to think about molecular bonds. The HCI molecule has a force constant k = 481 N/m. For the mass, use the reduced mass, which is defined as µ = (m₁m₂)/(m₁+m₂). a) Plot the potential energy of HCl from -1 to 1 Å. What happens to the curvature of the potential as the force constant is varied? What does this mean physically? b) Plot position as a function of time for a total energy of 6 x 10-20 J. What is the period of the motion? How does the period change as the force constant is varied? Explain why this makes sense physically.arrow_forward8.27 A paramagnetic ideal gas obeys Curie's law: Xm= a/T, where Xm is the susceptibility and a is a constant. A volume Vo of the gas is placed in a magnetic field of flux density Bo, which is then reduced adiabatically to zero. How must the volume be changed as a function of field if the temperature of the gas is to remain constant?arrow_forwardUse Avogadro’s constant to relate themass and molar mass of molecules andionic compounds to their elementaryentities (atoms or ions)arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

