
Concept explainers
Interpretation:
It should be explained that why two different products are formed from disrotatory ring closure of (2E, 4Z, 6Z)-octatriene, but only one product is formed from disrotatory ring closure of (2E, 4Z, 6E)-octatriene.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
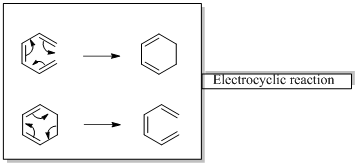
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
Enantiomers are a type of stereoisomers that have the same molecular formula and constitutions around the atom but differ in their spatial arrangement of groups around the atom.
Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry
- Draw the products (including the stereochemistry) formed in the following reaction.arrow_forwardDraw the products obtained (including stereochemistry) when each compound is treated with LDA, followed by CH3I.arrow_forwardTreatment of 2,2-dibromobutane with two equivalents of strong base affords but-1-yne and but-2-yne, as well as a small amount of buta-1,2-diene. Draw a mechanism showing how each compound is formed. Which alkyne should be the major product?arrow_forward
- List all alkene products for each reaction. Write a mechanism for the formation of each product following E1 or E2 mechanisms as appropriate. Be sure to draw arrows for the mechanism.arrow_forwardDetermine the mechanism of nucleophilic substitution of each reaction. Draw the products, including stereochemistry.arrow_forwardIdentify the product in each reaction, and explain why starting materials with identical functional groups give different products.arrow_forward
- Select the elimination product formed in this reaction. A B D с A E B B CI B NaOCH3 CH₂OH C D None of these is the E2 product Earrow_forwardDraw reaction mechanisms with all reactants, arrows, intermediates, and products. Your mechanism must account for all the products if more than one product is formed. 4-methycyclohexanol with phosphoric acid H3PO4 to for 1-methycyclohexene, 3- methylcyclohexene and 4-methycyclohexenearrow_forwardAs we will learn in Chapter 9, an epoxide is an ether with an oxygen atom in a three-membered ring. Epoxides can be made by intramolecular SN2 reactions of intermediates that contain a nucleophile and a leaving group on adjacent carbons, as shown.Assume that each of the following starting materials can be converted to an epoxide by this reaction. Draw the product formed (including stereochemistry) from each starting material. Why might some of these reactions be more difficult than others in yielding nucleophilic substitution products?arrow_forward
- Part A What is the major product formed when the following compound undergo an E1 reaction? CH, CH CH;CH,C–CHCH;CH3 Br for bonds ond chargee) Atoms gnd Terarrow_forwardDraw the keto and enol forms of 3,3-dimethylbutan-2-one, circle the more stable tautomer, and propose a mechanism for the conversion of one tautomer to the other in acidic conditions.arrow_forwardSelect the reagents that will allow the following transformation to occur. ? 1) О3 2) DMS OsO4 H2O2 mCPBA 1) ВН3 2)Н2О2, NaOH ОН ОН + enantiomerarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





