
Concept explainers
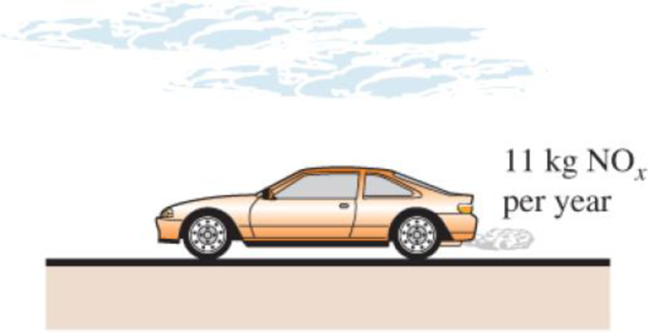
A typical car driven 20,000 km a year emits to the atmosphere about 11 kg per year of NOx (nitrogen oxides), which cause smog in major population areas. Natural gas burned in the furnace emits about 4.3 g of NOx per therm (1 therm = 105,500 kJ), and the electric power plants emit about 7.1 g of NOx per kWh of electricity produced. Consider a household that has two cars and consumes 9000 kWh of electricity and 1200 therms of natural gas per year. Determine the amount of NOx emission to the atmosphere per year for which this household is responsible.
FIGURE P2–83
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Thermodynamics: An Engineering Approach
Additional Engineering Textbook Solutions
EBK FUNDAMENTALS OF THERMODYNAMICS, ENH
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
DESIGN OF MACHINERY
Fundamentals Of Thermodynamics
Fluid Mechanics Fundamentals And Applications
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
- An engine uses about 0.24 kg of fuel/kW-hr. If the heating value of the fuel is 50 MJ/kg, what is the thermal efficiency?arrow_forwardA car engine with a power output of 65 hp has a thermal efficiency of 24%. Determine the fuel consumption rate of this car if the fuel has a heating value of 19,000 Btu/lb m. Answer ASAP thanks. I will rate helpfularrow_forwardKing Orquinaza bought a diesel electric plant in a remote part of Ilo-Ilo province that utilizes diesel fuel with an API of 28 at 15.6 deg C. The plant consumes 680 liters of diesel fuel at 26.6 deg C in 24 hours whie the power generated for the same period amounts to 1980 KW-hrs. Determine the density of the fuel at 26.6 deg C in kg/liters.arrow_forward
- A gas is contained in a vertical piston–cylinder assembly by a piston with a face area of 40 in2 and weight of 100 lbf. The atmosphere exerts a pressure of 14.7 lbf/in2 on top of the piston. A paddle wheel transfers 3 Btu of energy to the gas during a process in which the elevation of the piston increases. The change in internal energy of the gas is 2.12 Btu. The piston and cylinder are poor thermal conductors, and friction between them can be neglected. Determine the elevation increase of the piston, in ft.arrow_forwardA gasoline engine uses 0.01 kg/s of fuel while delivering 55 kW of power. The heating value of the fuel is 35,000 kJ/kg and the fuel density is 900 kg/m^3. In the question that follows, select the answer that is closest to the true value. What is the thermal efficiency of the engine?arrow_forwardA car traversing along Taft Avenue consumes 12 liters of gasoline per 100 kilometers. What amount of carbon dioxide is produced by the car?arrow_forward
- An automobile engine consumes fuel at a rate of 22 L/h and delivers 55 kW of power to thewheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determinethe efficiency of this engine. Hint, Qa= mass of fuel * heating value.arrow_forwardA steam power station uses coals of calorific value of 6045-kcal/kg. The overall efficiency ofthe power station is 21.5% and a coal power station uses 4.05-tonnes of coal every 15 minutesto generate the maximum demand. The purchasing price of coals is R600 per tonne. (NB:1 kWh = 861 kcal)(a) Determine the coal consumption in kg per kWh.(b) Determine the cost of purchasing the coals per week if the load factor is 65%.arrow_forwardOn a cold winter day, the rate of heat transfer from the air in a certain room is 5500 Btu/hr. The room is heated by a heating system, and the warm air is circulated by a fan which develops 0.75 hp. What is the required rate of heat transfer in kW, from the heating system in order to maintain constant room conditions?arrow_forward
- What is the thermodynamics parameter that refers to the total energy content of a system?arrow_forwardwhat is the term that refers to the total energy content of a system in the topic of Thermodynamics?arrow_forwardIn the United States, energy for household heating is generally sold using English units, e.g., therm, gal, and cord. A house in Wisconsin uses 1200 therms of thermal energy during the heating season. Calculate the cost of fuel if the furnace uses (a) natural gas with an efficiency of 70%; (b) No. 2 fuel oil, efficiency 65%; (c) kerosene, efficiency 99.9% (unvented); and (d) wood with l5% moisture with an efficiency of 50%. Use the data in Tables 2.2, 2.7, and 2.13. The efficiencies are based on the HHV. Assume the cost of natural gas is $8/MBtu, the cost of No. 2 fuel oil is $3/gal, the cost of kerosene is $3.50/gal, and the cost of wood is $100/cord. Assume the bulk density of cord wood is 30 lbm/ft3 .arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





