
Interpretation:
To identify the incorrect statement concerning the first step of the degradation of the heme portion of hemoglobin.
Concept introduction:
In the body, red blood cells deliver oxygen and remove carbon dioxide from the body tissues. Mature red blood cells have hemoglobin which is responsible for the oxygen carrying ability of the red blood cells.
Hemoglobin is a heme protein present in the red blood cells. The protein part is called globin and the non-protein part is heme. Heme is the prosthetic group that contains 4 pyrrole groups bonded together and has an iron atom in the center. The structure of the heme group is:
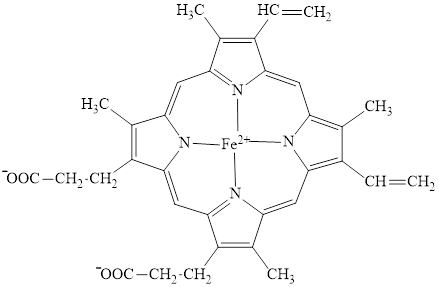
Red blood cells are formed in the bone marrow. The primary site for a breakdown of red blood cells is spleen and the secondary site is liver. Breakdown of red blood cells involves the degradation of hemoglobin. The protein part (globin) of the hemoglobin is hydrolyzed to an amino acid, an iron atom of the heme is converted to ferritin which is an iron storage protein and the remaining tetrapyrrole carbon arrangement upon degradation forms bile pigments.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
General, Organic, and Biological Chemistry
- Which of the following statements applies to a zymogen? a. inactive precursor of a proteolytic enzyme b. active form of a proteolytic enzyme c. substance that coverts an enzyme from an inactive form to an active form d. no correct responsearrow_forwardWhich of the following processes occurs within the cytosol of a cell? a. ketogenesis b. -oxidation pathway c. electron transport chain d. no correct responsearrow_forwardPractice Question • What is being oxidized in this reaction? - NADH + ADP + Phosphate + O2 + H+ → NAD+ + ATP + H,0 - (A) ADP (В) АТР (C) The phosphorus in ADP but not the whole molecule (D) NADHarrow_forward
- Which of the following hormones increases blood glucose levels? a. glucagon and epinephrine b. insulin and epinephrine c. insulin and glucagon d. more than one correct response e. no correct responsearrow_forward3. Which of the following chemical reactions is a nonredox reaction? HIDE ANSWER a. b. SO3 + H₂O → H₂SO4 C. Ca+S → CaS d. 2NO+ O2 → 2NO₂ no correct responsearrow_forward11. Phosphoglycerides are examples of a. bile salts b. phospholipids c. glycolipids d. waxes e. no correct responsearrow_forward
- 24. In the following diagram, the rate of an enzyme-catalyzed reaction is graphed on the y (or vertical) axis versus another quantity on the x axis. What quantity is graphed on the x axis? a. substrate concentration Ob. reaction pH Oc. reaction temperature Od. more than one response is correct e. no correct responsearrow_forward22. According to the lock-and-key theory of enzyme behavior, which of the following can be thought of as the key? a. enzyme b. substrate с. ароenzyme d. coenzyme e. no correct responsearrow_forwardExercise 24. Which of the following SN reactions will take place? 1. NH2 I NH2 + HO. + -F + OH 3. CN + CN + 2.arrow_forward
- 6. An enzyme without its cofactor is inactive. Another name for an inactive enzyme of this type is a. isoenzyme b. allosteric enzyme с. ароenzyme d. more than one response is correct e. no correct responsearrow_forwardWhich of the following polysaccharides contain both alpha 1 → 4 and beta 1 → 4 linkages? a. amylose b. cellulose c. amylopectin d. more than one choice is correct e. no correct response The cellular fluid in the interior of a cell is known as the a. cytoplasm b. matrix c. organelles d. lipid bilayerarrow_forwardR Review I Con MISSED THIS? Read Section 19.9 (Pages 878 - 881). Part A Under which set of conditions is AGrxn for the reaction A(g) B(g) most likely to be negative? PA = 70.0 atm; P = 0.070 atm %3D O PA = 0.070 atm; Pg = 0.070 atm %3D %3D O PA = 0.070 atm; PB = 70.0 atm O PA = 70.0 atm; PB = 70.0 atm Submit Request Answer Provide Feedbackarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

