
Concept explainers
(a)
Interpretation: To classify carbamoyl phosphate in terms of nitrogen content as
Concept introduction: Carbamoyl phosphate is an intermediate or one of the fuel for the urea cycle. Ammonium ion produced via oxidative deamination reaction is converted into the carbamoyl phosphate which then enters the urea cycle.
The carbamoyl phosphate formation reaction is carried out in the mitochondrial matrix. Carbamoyl is the prefix used to represent an amide group. Thus carbamoyl phosphate is a molecule that contains a phosphate group attached to an amide
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(a)
Answer to Problem 26.75EP
Carbamoyl phosphate is a
Explanation of Solution
Carbamoyl phosphate and aspartate are fuel for the urea cycle. The structure of carbamoyl phosphate is:
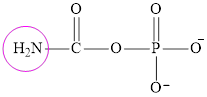
It contains only one nitrogen atom thus carbamoyl phosphate is characterized as
(b)
Interpretation: To classify glutamate in terms of nitrogen content as
Concept introduction: Glutamate is conjugate anion of glutamic acid. Glutamic acid is an amino acid. Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(b)
Answer to Problem 26.75EP
Glutamate is a
Explanation of Solution
The structure of glutamate is:
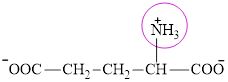
It contains only one nitrogen atom thus glutamate is characterized a
(c)
Interpretation: To characterize urea in terms of nitrogen content as
Concept introduction: The nitrogenous product of protein
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(c)
Answer to Problem 26.75EP
Urea is a
Explanation of Solution
The structure of urea is:
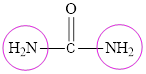
The structure of urea suggests the presence of two
(d)
Interpretation: To characterize citrulline in terms of nitrogen content as
Concept introduction: Citrulline is a nonstandard amino acid and is an inetrmediate in the the urea cycle. It is encountered at step 2 of the urea cycle and undergo condensation reaction with aspartate to give argininosuccinate.
Depending upon the number of nitrogen atom present in the structure of the compound it can be classified as
(d)
Answer to Problem 26.75EP
Citrulline is a
Explanation of Solution
The structure of citrulline is:
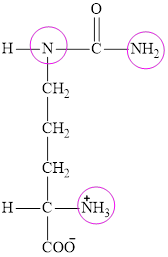
Citrulline contains three nitrogen atoms and thus it is characterized as a
Want to see more full solutions like this?
Chapter 26 Solutions
General, Organic, and Biological Chemistry
- (c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- reaction scheme for C39H4202 Hydrogenation of Alkyne (Alkyne to Alkene) show reaction (drawing) pleasearrow_forwardGive detailed mechanism Solution with explanation needed. Don't give Ai generated solutionarrow_forwardShow work with explanation needed....don't give Ai generated solutionarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





