
Interpretation:
The purine and pyrimidine ring structures should be drawn demonstrating the
Concept Introduction:
Heterocyclic
Explanation of Solution
Purine is represented in a double-ring structure which has four-nitrogen and five-carbon atoms. In this structure, carbon-2 and carbon-8 are obtained from N10 -formyl −H4folate and nitrogen-3 and nitrogen-9 are obtained from glutamine. There is no metabolic relation between purine and pyrimidines. Purine and Pyrimidines both have a different process for the synthesis and for the degradation. These both process of degradation and synthesis occur in all organisms. Structure of the purine is given below-
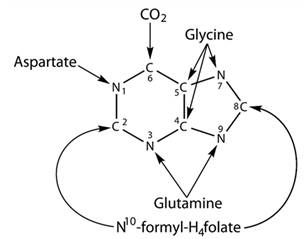
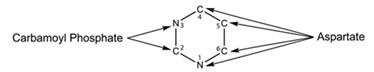
Pyrimidine is a six-membered single ring structure. It has two nitrogen and four carbon atoms. Nitrogen-3 and carbon-2 are obtained from carbamoyl phosphate. Carbon-4,5,6 and nitrogen-1 are obtained from aspartate. Structure for the pyrimidine is given below-
Want to see more full solutions like this?
Chapter 26 Solutions
Biochemistry
- Looking at the structure of alpha linolenic acid and knowing the electron carriers produced in each round of traditional beta oxidation for saturated fatty acid chains, how many rounds of beta oxidation will have diminished production of electron carriers due to the presence of double bonds?arrow_forwardThe four-carbon sugar erythrose-4-phosphate is the starting material in the synthesis of many compounds. Name two important examples.arrow_forwardIn many biochemical reactions which involves the formation of an enolate intermediate, the carbonyl oxygen of the substrate is coordinated to a divalent metal ion (usually zinc or magnesium) in the active site. Explain with structural drawings, how this ion-dipole interactions affect the acidity of the a-protons?arrow_forward
- Under certain conditions, peptide bond formation rather than peptide bond hydrolysis is thermodynamically favorable. Would you expect chymotrypsin to catalyze peptide bond formation? Explain.arrow_forwardWith regards to arganine illustrate the pH-dependent transitions on a titration graph from most to least protonated. Indicate also the relevant part of the amino acid which is associated with the transition on that part of the graph.arrow_forwardThe sedimentation value of aspartate transcarbamoylase decreases when the enzyme switches to the R state. On the basis of the allosteric properties of the enzyme, explain why the sedimentation value decreases.arrow_forward
- An inhibitor that specifically labels chymotrypsin at histidine 57 is Ntosylamido-l-phenylethyl chloromethyl ketone. How would you modify the structure of this inhibitor to label the active site of trypsin?arrow_forwardSerine protease enzyme mutation To show differences in the effect of the nucleophilic attack of the carbonyl group (C=O) of peptide bond between the catalytic triad of serine, histidine and aspartic acid, and another catalytic triad contains alanine, histidine and aspartic acid Provide/ draw an example of catalytic mechanism with catalytic triad contains alanine, histidine and aspartic Please answer completely will give rating surelyarrow_forwardSphingolipid storage diseases (sphingolipidoses) are inherited diseases caused by abnormal accumulation of membrane lipids. Elaborate on the molecular basis behind Tay-Sachs disease. Note: I expect a genuine explanation toward this question!arrow_forward
- a molecule that lowers cholesterol levels in humans interacts with the enzyme HMA-COG reductase. how do you describe their interaction biochemically? what experiment would you do?arrow_forwardthe following peptide is incubated as chymotrypsin: Gly-Val-Phe-Lys-Ala. Present the detailed chemical mechanism by which chymotrypsin hydrolyzes this peptide. Briefly explain each step. Include only the structure of the final products of the reaction.arrow_forwardAn inhibitor that specifically labels chymotrypsin at histidine 57 is N-tosylamido-l-phenylethyl chloromethylketone. How would you modify the structure of this inhibitor tolabel the active site of trypsin?arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
