
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Question
Chapter 2.5, Problem 11PTS
Interpretation Introduction
Interpretation: All the lone pairs associated to nitrogen atoms for the following compound should be drawn.
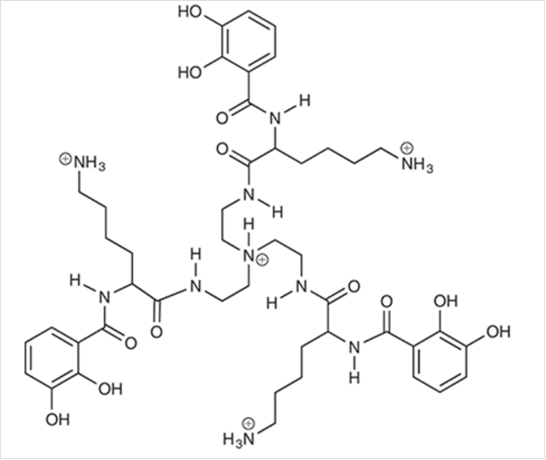
Concept Introduction The unshared pair of electrons are said to be lone pairs of electrons that are present in an atom of a compound.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Depending on the protein under study, the bond angle of a drug molecule can become critical to successfully deactivating a viral protein. For this reason, chemists are frequently concerned with the 3D shape of their molecules and their bond angles.
The oxygen atom shown in the structure has a tetrahedral electronic geometry, meaning we would predict that it would have 109.5° bond angles. However, the actual structure, the bond angles are smaller than 109.5°. Explain why this compression occurs. Make sure to discuss what’s happening around/what groups are present around the oxygen atom.
2. From the models of SF4, BrF3, and XeF4, deduce whether different atom arrangement, called
geometrical isomers, are possible; if so, sketch them below. Indicate the preferred geometry
for each case and suggest a reason for your choice. Indicate which structures have dipole
moments and show their direction,
Preferred
Molecular
Compound
Electron
Dipole
Reason
Geometry
Moment
(Polarity) geometry
SF4
BRF3
XeF4
3. Using the Lewis structure predict the geometrical structures of the following ions and state
the hybridization of the central atom.
Ion
Electron Geometry
Molecular
Formal Charge
Central Atom
Geometry
of Central
Hybridization
Atom
CO32-
NO3-
BF4
) Bean hexene is an odor compound used in cosmetics and cleaners. It is also known as leguminal. Its formula is:
CH3CH2CHCHCH2CH2OCHCH2CH3
|
OCH3
Please draw a Lewis structure for this compound with CORRECT GEOMETRY (use dashes and wedges!)
Label all pi bonds in the molecule (if there are any)
Indicate the bond angles for the red highlighted section of the molecule. (hint: you may want to keep the main carbon chain in-plane with the page for clarity)
For this question, you don’t need to draw out the orbitals.
Chapter 2 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 2.1 - Prob. 1LTSCh. 2.1 - Prob. 3ATSCh. 2.2 - Prob. 2LTSCh. 2.2 - Prob. 4PTSCh. 2.2 - Prob. 6ATSCh. 2.4 - Prob. 3LTSCh. 2.4 - Prob. 9ATSCh. 2.5 - Prob. 4LTSCh. 2.5 - Prob. 11PTSCh. 2.9 - Prob. 6LTS
Ch. 2.9 - Prob. 15PTSCh. 2.9 - Prob. 16PTSCh. 2.10 - Prob. 18CCCh. 2.10 - Prob. 19CCCh. 2.10 - Prob. 20CCCh. 2.10 - Prob. 21CCCh. 2.10 - Prob. 22CCCh. 2.10 - Prob. 23CCCh. 2.10 - Prob. 24CCCh. 2.10 - Prob. 25CCCh. 2 - Prob. 34PPCh. 2 - Prob. 35PPCh. 2 - Prob. 36PPCh. 2 - Prob. 37PPCh. 2 - Prob. 38PPCh. 2 - Prob. 40PPCh. 2 - Prob. 42PPCh. 2 - Prob. 43PPCh. 2 - Prob. 44PPCh. 2 - Prob. 45PPCh. 2 - Prob. 46PPCh. 2 - Prob. 47PPCh. 2 - Prob. 48PPCh. 2 - Prob. 49PPCh. 2 - Prob. 50PPCh. 2 - Prob. 67ASP
Knowledge Booster
Similar questions
- Vitamin B6 is an organic compound whose deficiency in the human body can cause apathy, irritability, and an increased susceptibility to infections. Below is an incomplete Lewis structure for vitamin B6. Complete the Lewis structure and answer the following questions. Hint: Vitamin B6 can be classified as an organic compound (a compound based on carbon atoms). The majority of Lewis structures for simple organic compounds have all atoms with a formal charge of zero. Therefore, add lone pairs and multiple bonds to the structure below to give each atom a formal charge of zero. a. How many bonds and bonds exist in vitamin B6? b. Give approximate values for the bond angles marked a through g in the structure. c. How many carbon atoms are sp2 hybridized? d. How many carbon, oxygen, and nitrogen atoms are sp3 hybridized? e. Does vitamin B6 exhibit delocalized bonding? Explain.arrow_forwardAspartame is an artificial sweetener marketed under the name Nutra-Sweet. A partial Lewis structure for aspartame is shown below. Aspartame can be classified as an organic compound (a compound based on carbon atoms). The majority of Lewis structures for simple organic compounds have all atoms with a formal charge of zero. Therefore, add lone pairs and multiple bonds to the structure above to give each atom a formal charge of zero when drawing the Lewis structure. Also note that the six-sided ring is shorthand notation for a benzene ring (C6H5). Benzene is discussed in Section 4-7. Complete the Lewis structure for aspartame. How many C and N atoms exhibit sp1 hybridization? How many C and O atoms exhibit sp3 hybridization? How many and bonds are in aspartame?arrow_forwardGlue adhesion usually requires a dry surface, so studying the chemistry behind the attachment of mussels to wet rocks can lead to the development of new adhesive materials. Chemists used the compound shown to model the mussel foot proteins involved in the adhesion process (Science 2015, 349, 628-632). Some of the nitrogen atoms in this structure are believed to play a critical role in achieving the strong binding. Identify the number of lone pairs on each nitrogen atom in the following structure. но но 8 H N° NH3 10 "Ne 6. タール OH 3. HO HO. 1 N: 6 N: 2 N: 7 N:arrow_forward
- How do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?arrow_forwardShould 100% sure of answer. Draw the Lewis structure of HBrO₃ (by following the octet rule on all atoms) and then choose the appropriate pair of molecular geometries of the two central atoms. Your answer choice is independent of the orientation of your drawn structure.arrow_forwardDesignate the structural features of polar molecules using partial charges or arrows. Ethyl heptanoate is responsible for grape aroma. It has the chemical formula C9H1802 but structural formula CH3-(CH2)s-COO-CH2-CH3. A structure is shown below. Which bonds would feature partial charges? Designate them using partial charges or arrows. H H H | H H H Harrow_forward
- Please envision this table like it was one long table. (I had to make it two because it hung out of the quiz boundaries and was inaccessible.) It is recommended that you draw the Lewis Structure of the molecules and then complete this table. electronic Molecular Molecule Geometry Geometry CH3F tetrahedral tetrahedral NH3 tetrahedral trigonal pyramidal C2F4 trigonal planar trigonal planar CO2 linear linear main dipole Molecule intermolecular moment force CH3F [ Select ] yes NH3 hydrogen bonding yes C2F4 [ Select ) no CO2 ( Select | no > >arrow_forwardAnswer the questions in the table below about the shape of the sulfur tetrafluoride (SFA) molecule. How many electron groups are around the central sulfur atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central sulfur atom? (You may need to use the scrollbar to see all the choices.) v (choose one) linear bent T-shaped trigonal planar trigonal pyramidal square planar square pyramidal tetrahedral sawhorse trigonal bipyramidal octahedralarrow_forward11 Name: Maddie Klink Instructor: Lab Day: Models of Organic Molecules Please bring your molecular model kits to lab for the completion of these exercises. Molecular Modeling: This lab is designed to help you become comfortable using model kits to visualize molecular structures in three dimensions. Part I: Answer these questions before doing any work with your model kit 1. How many bonds must each of the following atoms have in order to have no formal charge? 4 valence 1450na a. Carbon b. Hydrogen bond c. Oxygen d. Chlorine 7 valence e. Nitrogen 5 valence Octet H- H Complete octet = 8-4 H 8-S = 3 3bonds 2. Using the information from question #1, determine whether each of the following structures is possible or impossible? (Note that if no formal charges are shown, this implies that all formal charges are zero.) 6 valence Octet = 8.6 H H с-н H 01C C -K = 2 bonas 8-1-1 bond H H H-C Lab Time: H -C-H H H H-C-CI- C-H ++ H H -H HIC C-H H VEUR 1arrow_forward
- Answer the questions in the table below about the shape of the fluoroform (CHF3) molecule. How many electron groups are around the central carbon atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central carbon atom? (You may need to use the scrollbar to see all the choices.) 0 (choose one) X 8arrow_forwardDraw out Lewis Structures for CO2 and H2O.Using VSEPR Theory make models to represent the two molecules [you do not have to showdouble bonds in your model].From your answer predict the shape of CS2 and H2S. Looking at the Periodic Table predict which of the following molecules will have the sameshape. Using VSEPR theory determine what that shape will be.NCl3 PF3 AlCl3 BrF3arrow_forwardELECTRONIC STRUCTURE AND CHEMICAL BONDING Predicting deviations from ideal bond angles Consider the chlorite (C102) a anion. What is the central atom? Enter its chemical symbol. How many lone pairs are around the central atom? What is the ideal angle between the chlorine-oxygen bonds? Compared to the ideal angle, you would expect the actual angle between the chlorine-oxygen bonds to be... 0 0 (choose one) X 3 1/5arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning