
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 44P
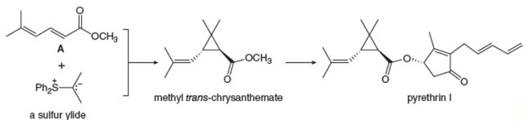
Sulfur ylides, like the phosphorus ylides of Chapter 21, are useful intermediates in
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Sulfur ylides, like the phosphorus ylides of Chapter 21, are useful intermediates in organic synthesis. Methyl trans-chrysanthemate, an intermediate in the synthesis of the insecticide pyrethrin I (Section 26.4), can be prepared from diene A and a sulfur ylide. Draw a stepwise mechanism for this reaction.
Electrophilic Addition
Soubong neblA-aleid rose 9160910 31 babeen ene singo
14.43 Draw the products formed when each compound is treated with one equivalent of HBr.
a.
b.
C.
One step in the synthesis of the antihistamine fexofenadine involves acid-catalyzed hydration of the triple bond in A. Draw a stepwise mechanism for this reaction and explain why only ketone B is formed.
Chapter 24 Solutions
Organic Chemistry (6th Edition)
Ch. 24.1 - Prob. 1PCh. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.2 - Prob. 4PCh. 24.2 - Prob. 5PCh. 24.4 - Problem 26.10
What reagents are needed to convert...Ch. 24.5 - Problem 26.11
What product is formed when each...Ch. 24.5 - Prob. 13PCh. 24.6 - Problem 26.13
Draw the products formed when each...Ch. 24.6 - Problem 26.14
What products are formed when ...
Ch. 24 - 26.19 What product is formed by ring-closing...Ch. 24 - 26.20 Draw the products formed in each...Ch. 24 - What organic halide is needed to convert lithium...Ch. 24 - 26.22 How can you convert ethynylcyclohexane to...Ch. 24 - 26.23 What compound is needed to convert styrene...Ch. 24 - 26.24 What steps are needed to convert to octane...Ch. 24 - Prob. 27PCh. 24 - 26.27 Draw the products (including stereoisomers)...Ch. 24 - 26.28 Treatment of cyclohexene with and forms...Ch. 24 - Prob. 32PCh. 24 - 26.30 What starting material is needed to prepare...Ch. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - When certain cycloalkenes are used in metathesis...Ch. 24 - 26.34 Draw the products formed in each reaction.
...Ch. 24 - Prob. 41PCh. 24 - Draw a stepwise mechanism for the following...Ch. 24 - Sulfur ylides, like the phosphorus ylides of...Ch. 24 - Although diazomethane is often not a useful...Ch. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - 26.45 Devise a synthesis of each compound from...Ch. 24 - 26.46 Devise a synthesis of each substituted...Ch. 24 - Biaryls, compounds containing two aromatic rings...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Devise a synthesis of compound A from the given starting materials. You may use any other inorganic reagents or organic alcohols. A was used to prepare aliskiren, a drug used to treat hypertension (see also Problem 5.7). CH,0 NH2 OH но. CHO Br CH,0. H2N. CH;0 CH30 A CH,0 Br Br aliskirenarrow_forwardLexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A. Draw a stepwise mechanism for this process.arrow_forwardDraw a stepwise mechanism for the following reaction, a key step in the synthesis of conivaptan (trade name Vaprisol), a drug used in the treatment of low sodium levearrow_forward
- Devise a synthesis of each compound from benzene. но OH CH3 а. b. с. d.arrow_forwardBromoetherication, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 21.37). Draw a stepwise mechanism for the following intramolecular bromoetherication reaction.arrow_forwardDevise a synthesis of each compound from cyclohexene as the startingmaterial. More than one step is needed.arrow_forward
- Devise a synthesis of each compound from aniline (C6H5NH2) as starting material.arrow_forwardDevise a synthesis of each compound from phenol (C6H5OH) and any other organic or inorganic reagents.arrow_forwardDraw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant uoxetine (trade name Prozac) in a single step.arrow_forward
- provide the missing reagents as appropriatearrow_forwardBromoetherification, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 18.34). Draw a stepwise mechanism for the following intramolecular bromoetherification reaction.arrow_forward16.41 Draw the products of each reaction. а. HNO3 H2SO4 'NO2 HO b. AICI3 CI С. H. Br2 FeBr3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY