
Concept explainers
(a)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
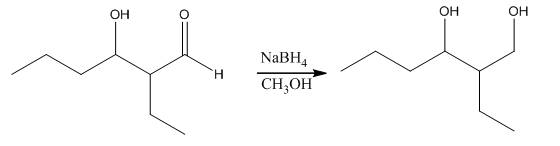
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown as,
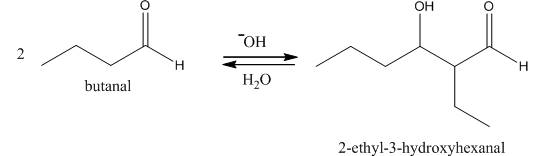
Figure 1
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 2
Thus, the reagent that is needed to convert
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(b)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
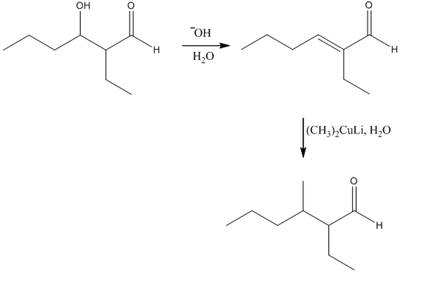
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
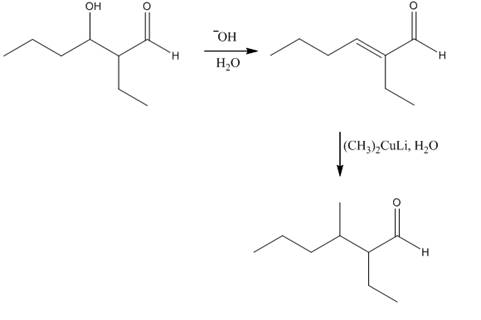
Figure 3
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(c)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
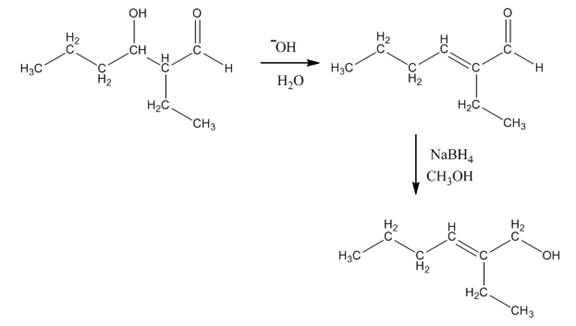
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert
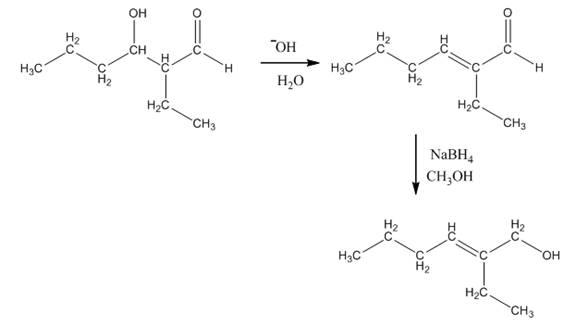
Figure 4
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
(d)
Interpretation: The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is to be shown. The reagents that are needed to convert the obtained product to the given compound are to be predicted.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.9P
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
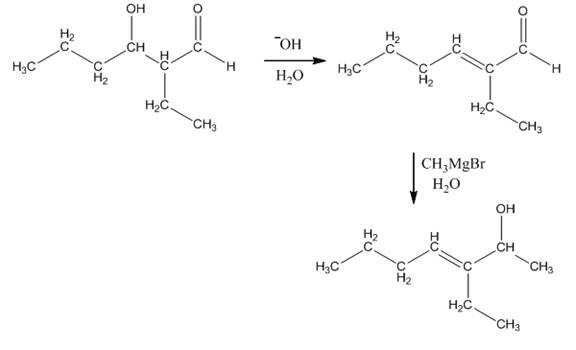
Explanation of Solution
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is shown in Figure 1.
In this reaction, first of all, one equivalent of butanal is treated with the strong base that results in the formation of a resonance-stabilized enolate ion. This enolate ion reacts with the second equivalent of butanal followed by the hydrolysis that leads to the formation of the desired product,
The reagents that are needed to convert

Figure 5
In this reaction,
The aldol product that is formed by the reaction of the two molecules of butanal in the presence of base is
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- draw the products obtained in each of the following reactionsarrow_forwardPlease draw substitution and elimination major productsarrow_forward16. Draw the structure of the aldol self-condensation (followed by dehydration) product for each of the following compounds. If a compound does not undergo aldol self-condensation, explain why it does not. a) || CH3CHCH₂CH T CH₂ b)arrow_forward
- Draw the product formed when A is treated with below series of reagents. [1] H2O; [2] NaH; [3] CH3Brarrow_forwardDraw the enol tautomers for each of the following compounds. For compounds that have more than one enol tautomer, indicate the one that is more stable.arrow_forwardDraw the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction followed by dehydration, when possible.arrow_forward
- Draw the main organic product formed in each of the following reactions:arrow_forwardWhat is the major product for each of the following reactions? (If an enol is created, write both the enol and keto products. If both ortho and para products are made, write both.)arrow_forwardWhat is the major substitution product of this reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

