
Draw the organic products formed when butanal
a.
b.
c.
d.
e.
(a)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
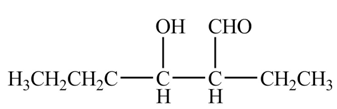
Explanation of Solution
The chemical equation when
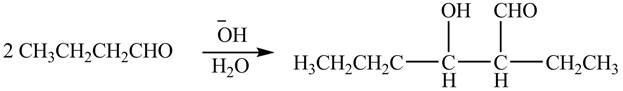
Figure 1
The above equation shows that two equivalents of butanal is treated with
The organic product formed when
(b)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
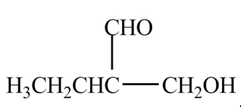
Explanation of Solution
The chemical equation when
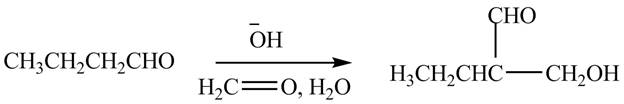
Figure 2
The above equation shows that butanal is treated with
The organic product formed when
(c)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
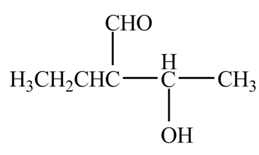
Explanation of Solution
The chemical equation when
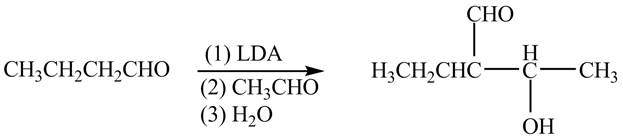
Figure 3
The above equation shows that butanal is treated with LDA,
The organic product formed when
(d)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
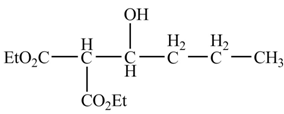
Explanation of Solution
The chemical equation when
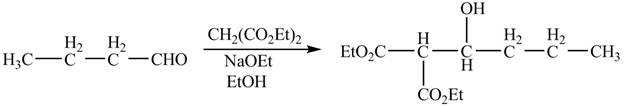
Figure 4
The above equation shows that butanal is treated with
The organic product formed when
(e)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
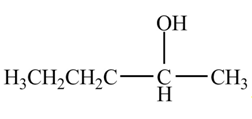
Explanation of Solution
The chemical equation when
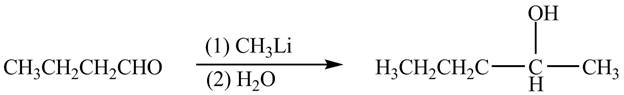
Figure 5
The above equation shows that butanal is treated with
The organic product formed when
(f)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
Explanation of Solution
The chemical equation when
The above equation shows that butanal is treated with
The organic product formed when
(g)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product does not form when butanal is treated with the given reagent.
Explanation of Solution
The chemical equation when
The above equation shows that butanal does not form any product when it is treated with hydrogen in the presence of
The reaction does not take place when butanal is treated with the given reagent.
(h)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
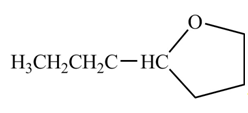
Explanation of Solution
The chemical equation when
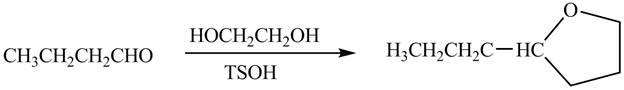
Figure 6
The above equation shows that butanal is treated with
The organic product formed when
(i)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
Explanation of Solution
The chemical equation when
The above equation shows that butanalreacts with
The organic product formed when
(j)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
Explanation of Solution
The chemical equation when
The above equation shows that butanal reacts with
The organic product formed when
(k)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
Explanation of Solution
The chemical equation when
The above equation shows that butanal reacts with
The organic product formed when
(l)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
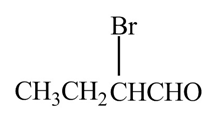
Explanation of Solution
The chemical equation when
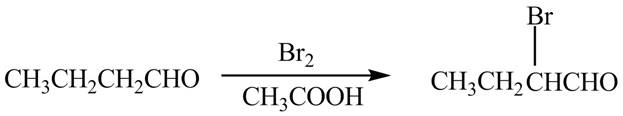
Figure 7
The above equation shows that butanal reacts with
The organic product formed when
(m)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
Explanation of Solution
The chemical equation when
Figure 8
The above equation shows that butanal reacts with
The organic product formed when
(n)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
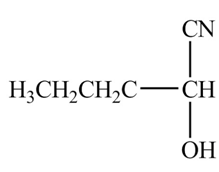
Explanation of Solution
The chemical equation when
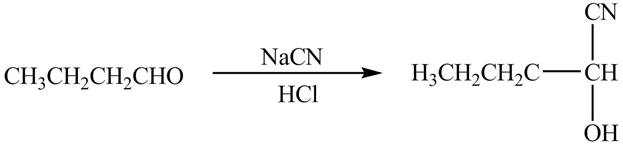
Figure 8
The above equation shows that butanal reacts with
The organic product formed when
(o)
Interpretation: The organic product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of products with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 24.48P
The organic product formed when
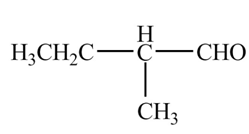
Explanation of Solution
The chemical equation when
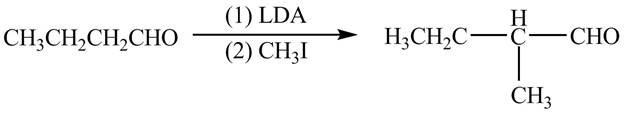
Figure 9
The above equation shows that butanal reacts with LDA in the presence of
The organic product formed when
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- OH 15. CH3-C-H ÓCH, The compound above is an example of a(n) a. acetal b. ketal O c. hemiacetal d. hemiketal e. none of the abovearrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forwardGive the products of the following acid–base reactions and indicate whether reactants or products are favored at equilibrium. a.CH3COH + CH3O− b. CH3CH2OH + −NH2 c. CH3COH + CH3NH2 d. CH3CH2OH + HClarrow_forward
- Classify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forwardWhat is the major organic product obtained from the following reaction? A. CI B. H 1. (CH3CH₂)2CuLi 2. H3O+ C. D. •ay.a OHarrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardDraw the structure of a dihalide that could be used to prepare each alkyne. There may be more than one possible dihalide. CH3 b. CH₂-C-C=CH CH₂ a. CH₂C=CCH₂ C. -C=C-arrow_forwardWhat is the major organic product obtained from the following reaction? A. B. OH COOH CI 1. Mg 2. CO2 3. H3O+ C. D. CH₂OH CHOarrow_forward
- Draw the product formed when pentanal (CH;CH,CH2CH;CHO) is treated with each reagent. With some reagents, no reaction occurs. a. NaBH4, CH,OH b. [1] LIAIH4; (2] H20 c. H2, Pd-C d. PCC e. NazCr,07, H2SO4, H2O f. Ag,0, NH,OH g. [1) CH,MgBr; (2] H2O h. [1] CęH,Li; [2] H20 i. [1] (CH3),CuLi; (2] H2O j. [1] HC=CNa; [2] H2O k. [1] CH;C=CLi; [2] H2O I. The product in (a), then TBDMS-CI, imidazolearrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardInstructions: Draw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a carboxylic acid, ester, or amide. a. CH3CH2CO2CH2CH3 b. CH3CONHCH3 c. (CH3)3CCO2H d. (CH3)2CHCON(CH3)2 Instructions: Give the IUPAC name for each compound. A. CH₂ CH₂CH₂CH₂CCH₂COOH CH3 B. CH₂CHCH₂CH₂COOH CH₂COOH CH₂CH3 C. (CH,CH,),CHCH,CHCOOH Instructions: Give the structure corresponding to each IUPAC name. a. 2-bromobutanoic acid b. 2,3-dimethylpentanoic acid c. 2-ethyl-5,5-dimethyloctanoic acid d. 3,4,5,6-tetraethyldecanoic acidarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
