
Concept explainers
(a)
Interpretation:
Whether hexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an
The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The sugar hexose is not the correct description of
Explanation of Solution
Monosaccharide with the six-carbon atom is known as hexose.
The presence of a keto group in
However, the keto group is missing in hexose as shown below.
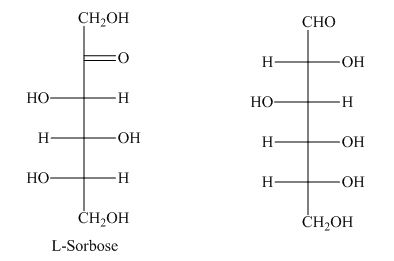
Figure 1
The term hexose is not the correct description of the
(b)
Interpretation:
Whether ketohexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The sugar ketohexose is the correct description of
Explanation of Solution
The class of the sugars which is fundamental in carbohydrates with presence of keto group is known as ketohexose sugars.
The presence of keto group in ketohexose shows similarity with
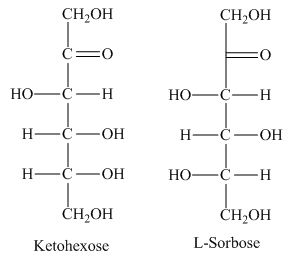
Figure 2
The ketohexose is the correct description of the
(c)
Interpretation:
Whether glycoside molecule corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The carbohydrate
Explanation of Solution
Two molecules of sugar which are connected to a glycosidic bond is known as glycoside molecule. The glycosidic bond is used to join two carbohydrate molecules.
The cyclic acetal group is not present in the
The glycoside molecule is not the correct description of the
(d)
Interpretation:
Whether aldohexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The carbohydrate
Explanation of Solution
The aldohexose belongs to the category of hexose in which aldehyde group is present at first carbon. In the
The sugar
(e)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is the correct description of
Explanation of Solution
The stereocenters are defined as the centers which are chiral in nature or attached with four different substituents. The stereocenters which are in direction of anticlockwise or left-hand nomenclaturehave
The configuration of
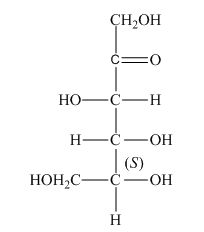
Figure 3
Therefore, the given structure is the correct description of
The given structure shown in Figure 3 is the correct description of
(f)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The stereocenters are defined as the centers which are chiral in nature or attached with four different substituents. The stereocenters which are in the direction of anticlockwise or left-hand nomenclature, the configuration of that stereocenter is
The configuration of
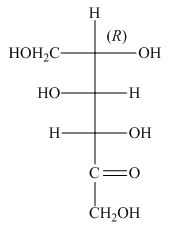
Figure 4
The given structure shown in Figure 4 is not the correct description of
(g)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
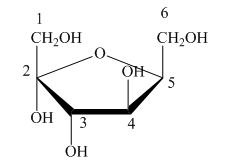
Figure 5
The given structure is not a proper description for
(h)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
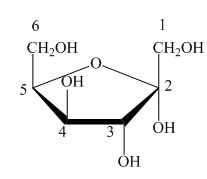
Figure 6
The configuration of sorbose is in
(i)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
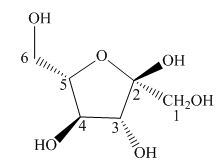
Figure 7
The given structure is the correct description for
(j)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
The pyranoses can be classified as
Answer to Problem 24.40AP
The given structure is not the correct description of
Explanation of Solution
The most stable conformation of cyclohexane is chair form due to the axial and equatorial position. The angle between the carbon-carbon bond is near about
The given structure is not the correct description of
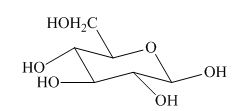
Figure 8
The given structure is not the correct description of
(k)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is the correct description of
Explanation of Solution
The most stable conformation of cyclohexane is chair form due to the axial and equatorial position. The angle between the carbon-carbon bond is near about
All the substituents or groups are present in the same manner with respect to the
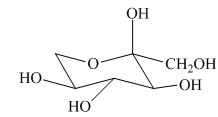
Figure 9
The given structure is the correct description of
Want to see more full solutions like this?
Chapter 24 Solutions
EBK ORGANIC CHEMISTRY
- 5. Provide suitable responses for questions (a) – (). 6 CH2OH 4 OH OH 3 OH (a) What is the relative configuration of the above monosaccharide? (b) Which labeled carbon is the anomeric carbon? (c) Trace and identify the acetal in the above monosaccharide. (d) Draw the hemiacetal that results from above acetal. (e) Draw the open chain equivalent of the sugar in part (d). (f) Classify the monosaccharide below as a D-sugar or an L-sugar. H. OH O. OH CH,OH OH OHarrow_forwardThe most stable conformation of the pyranose ring of most Daldohexosesplaces the largest group, CH2OH, in the equatorial position.An exception to this is the aldohexose D-idose. Draw the two possiblechair conformations of either the α or β anomer of D-idose. Explain whythe more stable conformation has the CH2OH group in the axial position .arrow_forwardThe most stable conformation of the pyranose ring of most D-aldohexoses places the largest group, CH2OH, in the equatorial position. An exception to this is the aldohexose D-idose. Draw the two possible chair conformations of either the a or B anomer of D-idose. Explain why the more stable conformation has the CH2OH group in the axial position.arrow_forward
- (a) Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b) The alcohol or phenol formed from the hydrolysis of a glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?arrow_forwardWhen a pyranose is in the chair conformation in which the CH2OH group and the C-1 OH group are both in axial positions, the two groups can react to form an acetal. This is called the anhydro form of the sugar (it has “lost water”). The anhydro form of D-idose is shown here. Explain why about 80% of d-idose exists in the anhydro form in an aqueous solution at 100 °C, but only about 0.1% of D-glucose exists in the anhydro form under the same conditions.arrow_forwardWhen a pyranose is in the chair conformation in which the CH2OH group and the C-1 OH group are both in axial positions, the two groups can react toform an acetal. This is called the anhydro form of the sugar (it has “lost water”). The anhydro form of d-idose is shown here. Explain why about 80% of d-idose exists in the anhydro form in an aqueous solution at 100 °C, but only about 0.1% of d-glucose exists in the anhydro form under the same conditions.arrow_forward
- 31. Which of the following statements about cholesterol is not correct? CH. HO Cholesterol 16 (a) Cholesterol is a steroid that contains a tetracyclic ring system. (b) Cholesterol is a steroid that contains 8 chiral carbons and can form 28 or 256 stereoisomers. (c) Each atom or group attached to a ring-junction carbon (i.e., carbons a-e) is in a trans or axial position. Because of this the tetracyclic ring system is mostly flat. (d) Cholesterol is used to synthesized vitamin D, bile acids, sex hormones, and adrenocorticoid hormones. (e) Cholesterol is not found in the cell membranes of animals.arrow_forwardBelow are drawn the different chair conformations of the methyl glycoside of galactose. но он но он оМе OMe но- в A OH он (ii) Give the correct full name of compound A (= B) (ii) The name of the compound does not contain information on conformation (only configuration). Give the accepted conformational description for compound A and for compound B. (iv) Draw the Fischer projections of the open form of the corresponding reducing sugars of compound A (= B).arrow_forward(i) Acetylation of glucose with acetic anhydride gives glucose pentacetate. Write the structure of the pentacetate. (ii) Explain – Why glucose pentacetate does not react with hydroxylamine?arrow_forward
- Consider the structure of raffinose, a trisaccharide found in sugar beets and a number of higher plants. HO CH,OH Но- OH OCH, Но Но OH НОСН Но CH,OH ÓH raffinose (a) Classify raffinose as a reducing or nonreducing sugar, and tell how you know. (b) Identify the glycoside linkages in raffinose, and clas- sify each as either a or B. (c) Name the monosaccharides formed when raffinose is hydrolyzed in aqueous acid. (d) What products are formed when raffinose is treated with dimethyl sulfate in NaOH, and then with aqueous acid and heat?arrow_forwardProvide suitable responses for questions (a) –(j).| 6 CH2OH 4 ОН OH OH 2.arrow_forwardGiven the following structure of aldose, (a) how many chiral carbons are there? (b) is it a reducing sugar? and (c) is it an L or D sugar?arrow_forward
