
Concept explainers
Interpretation:
In the nitration of phenol (
Concept introduction:
The substituent on the benzene ring prior to substitution dictates the regiochemistry of the reaction. Some substituents are designated as ortho/para directors because they lead to product mixtures consisting primarily of the ortho- and para-disubstituted products. Other substituents are designated as meta directors because they favor the formation of the meta-disubstituted product. The results from nitration reactions are summarized in Table 23-1:
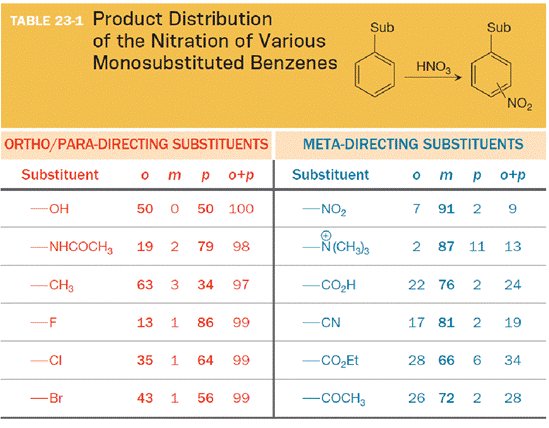
In the nitration of phenol (
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- What is the strongest nucleophile, NH3, CH4, CH3O-, or H2O?arrow_forwardGoing down a group, consider the halides: Of the halides shown, which is most basic? Of the halides shown, which is most nucleophilic? Br Of the halides shown, which is least basic? Of the halides shown, which is least nucleophilic?arrow_forwardGiven the general reaction coordinate diagram for an El reaction, what does the 1st transition state refer to? TS E TS, SM Reaction Coordinate An activated complex between the leaving group and carbocationic species. An activated complex between the nucleophile and carbocationic species. None of these O An activated complex between the base and carbocationic species.arrow_forward
- in the reaction above, how would the reaction shift (left or right) if we added extra NO2 to the reaction? What would the reaction color be after the shift?arrow_forwardDraw reaction of an a,b-unsaturated aldehyde or a ketone with a nucleophile forms a direct addition product and/or a conjugate addition product, depending on the strength of the nucleophile and the structure of the aldehyde or ketone.arrow_forwardIn the reaction CH₂O + HCN CH₂CNOH, which species serves as a nucleophile?arrow_forward
- Which of the following will react faster and whyarrow_forward4. Draw the reaction mechanism and product for the following reaction. Same HO [H*] Quyarrow_forwardIf the anionic species shown were to eliminate a leaving group, the three possi- bilities would be H3C¯, CI, or CH30¯. Draw the curved arrow notation and the products for each of these elimination steps. Which is the major product? Why? CIarrow_forward
- The reaction shown here proceeds via a carbocation rearrangement. Draw a complete, detailed mechanism to account for the product. Explain why the carbocation rearrangement is favorable. CH;OH Brarrow_forwardDraw curved arrows for the following reactionsarrow_forwardDraw the mechanism for this reaction. HO H,SO4.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





