
(a)
Interpretation:
Mechanism for the conversion of compound (A) to compound (B) has to be given.
Concept Introduction:
Acid catalyzed reactions:
In an
(b)
Interpretation:
For the conversion of compound (B) to compound (C) reagents has to be proposed.
Concept Introduction:
Grignard addition:
It is an addition of organomagnesium halide to an
(c)
Interpretation:
Mechanism for the conversion of compound (C) to compound (D) has to be given.
Concept Introduction:
Acid catalyzed reactions:
In an organic synthesis reactions, most of the reactions are initiated by the abstraction of proton from acids by the reactant. Sulphuric acid is one of the reagent used for this purposes.
(d)
Interpretation:
For the conversion of compound (d) to amitriptyline reagents has to be proposed.
Concept Introduction:
SN2 reaction:
SN2 stands for bimolecular nucleophilic substitution reaction. In this type of reactions, simultaneous bond breaking and bond making steps are involved.
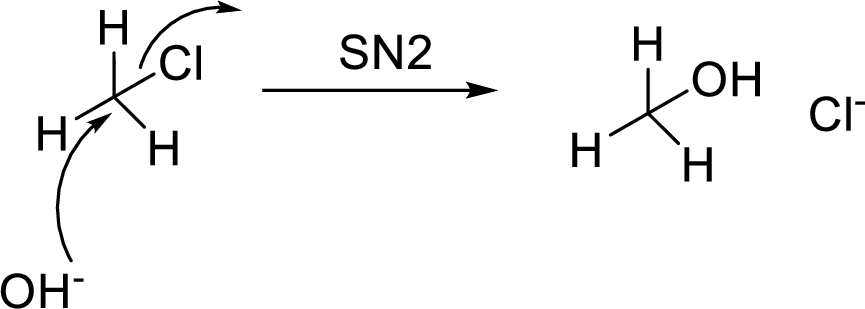
(e)
Interpretation:
The chirality of amitriptyline and the number of possible stereoisomers has to be checked.
Concept Introduction:
Stereoisomers and chiral:
Isomers differing only in their spatial arrangement of atoms are called as stereoisomers.
The geometric property of a molecule that cannot be superimposed on its mirror image is known as chirality. Chiral carbon is the one which is attached with four different groups or atoms.
Trending nowThis is a popular solution!

Chapter 22 Solutions
Organic Chemistry
- Paroxetine (Paxil) is an antidepressant that is a member of a family of drugs known as Selective Serotonin Reuptake Inhibitors (SSRIS). This family of drugs also includes fluoxetine (Prozac) and sertraline (Zoloft). SSRIS work by inhibiting the reuptake of the neurotransmitter serotonin in the synapses of the central nervous system follow- ing release of serotonin during excitation of individual nerve cells. Between firings, the serotonin is taken back up by a nerve cell in preparation for firing again. Inhibition of reuptake has the effect of increasing the time serotonin molecules remain in the syn- apses following excitation, leading to a therapeutic effect. In one synthesis of parox- etine, the following reagents are used. Draw the structures of synthetic intermediates A and B. F НО SOCI, A B HO Pyridinearrow_forwardWhich of the following is a meta-hydroxylated arylimidazoline alpha-1 direct-acting agonist? Naphazoline Oxymetazoline Phenylephrine Tetrahydrozolinearrow_forwardMechanism of drug action is explored by: * pharmacoeconomics pharmacodynamics O pharmacogenetics O pharmacokineticsarrow_forward
- What is the mechanism of action of the antibiotic drug Sulfanilamide in terms of enzyme inhibition?arrow_forwardWrite the therapeutic action of following on human body and mention the class of drugs to which each of the these belong:(i) Equanil(ii) Aspirin(iii) Chloramphenicolarrow_forwardWhat are the contraindications of norepinephrine as a drug?arrow_forward
- Bisphenol A is widely used as a building block in polymer synthesis and is found in the polycarbonate hard plastics of reusable drink containers, DVDs, cell phones, and other consumer goods. Bisphenol A is reported to have estrogenic activity, and its widespread occurrence in our environment is a potential concern. Describe one or two biochemical experiments that could be done to compare the activity of bisphenol A with that of its estradiol, its structural relative.arrow_forwardAspirin reduces the inflammation and fever through which of the following possible mechanism of action? Delay the secretion of the local hormone Leukotriene Preventing the promotion of thromboxanes in blood vessels Inhibit enzymes leading to prostaglandin synthesis Promotion of various inflammatory and hypersensitivity reactionsarrow_forwardExplain the biosynthetic pathway of caffeine to Theobromine in detail?arrow_forward
- ) The potency of antagonists is frequently defined in terms of either their IC50, ortheir Ki. Explain what these two terms mean.arrow_forwardWhy can some vitamin-K antagonists act as anticoagulants?arrow_forward1. (a) There are two main types of adrenergic receptors: the a- and B-adrenoceptors. Noradrenaline shows slight selectivity for the a-adrenoceptor, whereas isoprenaline shows selectivity for the B-adrenoceptor. Adrenaline shows no selectivity and binds equally well to both the a- and B-adrenoceptors. Suggest an explanation for these differences in selectivity. OH OH OH Но. NH2 HO. НО. НО Но Но Noradrenaline Isoprenaline Adrenaline (b) Isoprenaline undergoes metabolism to give the inactive metabolite shown below. Suggest why this metabolite does not bind to the B-adrenoceptor. OH MeO. Но inactive metabolite of Isoprenaline (c) R-Salbutamol is an anti-asthmatic agent that acts as an adrenergic agonist. Do you think it is likely to show any selectivity between the a- and B-adrenoceptors? Explain your answer. OH HOH2C, Но R-Salbutamolarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

