
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.23P
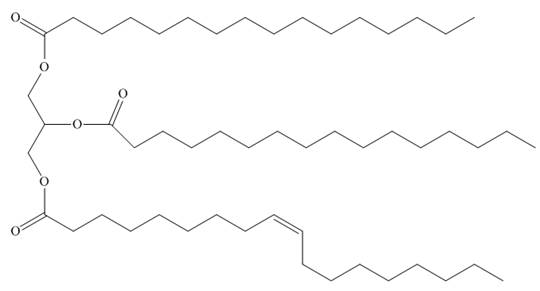
What is the composition of the soap prepared by hydrolysis of the following triacylglycerol?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used Ai solution
Don't used Ai solution
Indicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are false
Chapter 22 Solutions
Organic Chemistry
Ch. 22 - Prob. 22.1PCh. 22 - Draw the three possible resonance structures for...Ch. 22 - Prob. 22.3PCh. 22 - Give an IUPAC or common name for each compound. a....Ch. 22 - Problem 22.5 Draw the structure corresponding to...Ch. 22 - Problem 22.6 Explain why the boiling point of is...Ch. 22 - Problem 22.7 How would the compounds in each pair...Ch. 22 - Problem 22.8 Deduce the structures of compounds ...Ch. 22 - Problem 22.9 Without reading ahead in Chapter 22,...Ch. 22 - Rank the compounds in each group in order of...
Ch. 22 - Prob. 22.11PCh. 22 - Prob. 22.12PCh. 22 - Prob. 22.13PCh. 22 - Prob. 22.14PCh. 22 - Problem 22.15 Draw the products of each...Ch. 22 - Problem 22.16 Draw the products of each reaction.
...Ch. 22 - Prob. 22.17PCh. 22 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.19PCh. 22 - Problem 22.20 Fenofibrate is a...Ch. 22 - Problem 22.21 What product is formed when the...Ch. 22 - How would you synthesize olestra from sucrose?
Ch. 22 - Problem 22.23 What is the composition of the soap...Ch. 22 - Problem 22.24 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.25PCh. 22 - Problem 22.26 Some penicillins cannot be...Ch. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Problem 22.30 Glucosamine is a dietry supplement...Ch. 22 - Draw the products of each reaction. a. c. b.Ch. 22 - Draw a tautomer of each compound.
a. b. c.
Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Prob. 22.35PCh. 22 - Problem 22.36 Outline two different ways that can...Ch. 22 - 22.37 Rank the following compounds in order of...Ch. 22 - Prob. 22.38PCh. 22 - Prob. 22.39PCh. 22 - 22.40 Give the IUPAC or common name for each...Ch. 22 - 22.41 Give the structure corresponding to each...Ch. 22 - Prob. 22.42PCh. 22 - 22.43 Explain why is a stronger acid and a weaker...Ch. 22 - (a) Propose an explanation for the difference in...Ch. 22 - Draw the product formed when phenylacetic acid is...Ch. 22 - Draw the product formed when phenylacetonitrile ...Ch. 22 - 22.47 Draw the organic products formed in each...Ch. 22 - Prob. 22.48PCh. 22 - Prob. 22.49PCh. 22 - Prob. 22.50PCh. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - 22.54 Draw a stepwise mechanism f or the following...Ch. 22 - 22.55 When acetic acid () is treated with a trace...Ch. 22 - Prob. 22.56PCh. 22 - Prob. 22.57PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - 22.63 Acid-catalyzed hydrolysis of forms compound...Ch. 22 - 22.64 What carboxylic acid and alcohol are needed...Ch. 22 - Problem 22.65 Devise a synthesis of each compound...Ch. 22 - Prob. 22.66PCh. 22 - Prob. 22.67PCh. 22 - Prob. 22.68PCh. 22 - Prob. 22.69PCh. 22 - 22.70 What polyester or poly amide can be prepared...Ch. 22 - 22.71 What two monomers are needed to prepare each...Ch. 22 - Prob. 22.72PCh. 22 - 22.73 How can IR spectroscopy be used to...Ch. 22 - 22.74 Rank the following compounds in order of...Ch. 22 - 22.75 Identify the structures of each compound...Ch. 22 - 22.76 Identify the structures of A and B, isomers...Ch. 22 - Prob. 22.77PCh. 22 - 22.78 Identify the structure of compound C...Ch. 22 - 22.79 Identify the structures of D and E, isomers...Ch. 22 - 22.80 With reference to amides A and B, the...Ch. 22 - Prob. 22.81PCh. 22 - Prob. 22.82PCh. 22 - Prob. 22.83PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Draw a stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward(c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY