
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
4th Edition
ISBN: 9780134110684
Author: Randall D. Knight (Professor Emeritus)
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 21, Problem 38EAP
Prove that the work done in an adiabatic process i f is
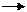 Ws= ( pfVf- piVi) / ( 1 - ? )
Ws= ( pfVf- piVi) / ( 1 - ? )
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
One kilogram of water at 0
C is heated to 100
C. Compute its change in entropy. Assume that the specific heat of water is constant at 4190 J/kg . K over this temperature range.
A large electrical power station generates 1000 MW of electricity with an efficiency of 35.0%. (a) Calculate the heat transfer to the power station, Qh, in one day. (b) How much heat transfer Qc occurs to the environment in one day? (c) If the heat transfer in the cooling towers is from 35.0C water into the local air mass, which increases in temperature from 18.0C to 20.0C, what is the total increase in entropy due to this heat transfer? (d) How much energy becomes unavailable to do work because of this increase in entropy, assuming an 18.0C lowest temperature? (Part of Qc could be utilized to operate heat engines or for simply heating the surroundings, but it rarely is.)
ft-lb
A certain ideal has a constant
gas
R= 38.9
Ib-R
with k= 1·4. Find the specific heat at constant volume
R.
if Cv can be computed by 78 t(k) Also, compute
BTU
for the specific heat at constant pressure.
Chapter 21 Solutions
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Ch. 21 - Prob. 1CQCh. 21 - Rank in order, from largest to smallest, the...Ch. 21 - Prob. 3CQCh. 21 - FIGURE Q21.4 shows the pV diagram of a heat...Ch. 21 - Rank in order, from largest to smallest, the...Ch. 21 - FIGURE Q21.6 shows the thermodynamic cycles of two...Ch. 21 - A heat engine satisfies Wout= Qnet. Why is there...Ch. 21 - Prob. 8CQCh. 21 - Prob. 9CQCh. 21 - Prob. 10CQ
Ch. 21 - Prob. 11CQCh. 21 - Prob. 1EAPCh. 21 - Prob. 2EAPCh. 21 - Prob. 3EAPCh. 21 - Prob. 4EAPCh. 21 - Prob. 5EAPCh. 21 - Prob. 6EAPCh. 21 - The power output of a car engine running at 2400...Ch. 21 - Prob. 8EAPCh. 21 - Prob. 9EAPCh. 21 - Prob. 10EAPCh. 21 - Prob. 11EAPCh. 21 - Prob. 12EAPCh. 21 - Prob. 13EAPCh. 21 - Prob. 14EAPCh. 21 - Prob. 15EAPCh. 21 - Prob. 16EAPCh. 21 - A heat engine uses a diatomic gas in a Brayton...Ch. 21 - At what pressure ratio does a Brayton cycle using...Ch. 21 - Prob. 19EAPCh. 21 - Prob. 20EAPCh. 21 - Prob. 21EAPCh. 21 - Prob. 22EAPCh. 21 - Prob. 23EAPCh. 21 - Prob. 24EAPCh. 21 - Prob. 25EAPCh. 21 - Prob. 26EAPCh. 21 - Prob. 27EAPCh. 21 - A Carnot engine whose hot-reservoir temperature is...Ch. 21 - Prob. 29EAPCh. 21 - A heat engine operating between energy reservoirs...Ch. 21 - Prob. 31EAPCh. 21 - A Carnot refrigerator operating between —20°C and...Ch. 21 - The coefficient of performance of a refrigerator...Ch. 21 - A Carnot heat engine with thermal efficiency 1/3...Ch. 21 - Prob. 35EAPCh. 21 - Prob. 36EAPCh. 21 - A heat engine with 50% of the Carnot efficiency...Ch. 21 - Prove that the work done in an adiabatic process i...Ch. 21 - Prob. 39EAPCh. 21 - Prob. 40EAPCh. 21 - An ideal refrigerator utilizes a Carnot cycle...Ch. 21 - Prob. 42EAPCh. 21 - There has long been an interest in using the vast...Ch. 21 - A Carnot heat engine operates between reservoirs...Ch. 21 - A Carnot engine operates between temperatures of...Ch. 21 - Prob. 46EAPCh. 21 - A Carnot heat engine and an ordinary refrigerator...Ch. 21 - 48. A heat engine running backward is called a...Ch. 21 - 49. A car's internal combustion engine can be...Ch. 21 - Prob. 50EAPCh. 21 - Prob. 51EAPCh. 21 - Prob. 52EAPCh. 21 - Prob. 53EAPCh. 21 - Prob. 54EAPCh. 21 - Prob. 55EAPCh. 21 - Prob. 56EAPCh. 21 - Prob. 57EAPCh. 21 - A heat engine using a monatomic gas follows the...Ch. 21 - Prob. 59EAPCh. 21 - Prob. 60EAPCh. 21 - Prob. 61EAPCh. 21 - Prob. 62EAPCh. 21 - Prob. 63EAPCh. 21 - Prob. 64EAPCh. 21 - Prob. 65EAPCh. 21 - Prob. 66EAPCh. 21 - Prob. 67EAPCh. 21 - Prob. 68EAPCh. 21 - Prob. 69EAPCh. 21 - Prob. 70EAPCh. 21 - A refrigerator using helium gas operates on the...Ch. 21 - Prob. 72EAPCh. 21 - The gasoline engine in your car can be modeled as...Ch. 21 - Prob. 74EAP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A dilute gas expands quasi-statically to three times its initial volume. Is the final gas pressure greater for an isothermal or an adiabatic expansion? Does your answer depend on whether the gas is monatomic, diatomic, or polyatomic?arrow_forwardProblem 9.00 SI At the beginning of the compression process of an air-standard Otto cycle, P - 1 bar and T, - 300 K. The compression ratio is 8.5 and the heat addition per unit mass of air is 1400 k/kg. Determine: (a) the maximum temperature of the cycle, in K. (b) the net work, in k/kg. (c) the percent thermal efficiency of the cycle. (d) the mean effective pressure, in kPa. v Part A Determine the maximum temperature of the cycle, in K. Tmax = the tolerance is +/-2% Click if you would like to Show Work for this question: Open Show Work By accessing this Question Assistance, you will learn while you cam points based on the Point Potential Policy set by your instructor. Attempts: 0 of 1 used SAVE FOR LATER SUBHIT ANSWER Earn Maximum Points available only if you answer this question correctly in your first attempt. Part B The parts of this question must be completed in order. This part will be avaliable when you complete the part above. Part C The parts of this question must be completed…arrow_forwardEn: The latent heat of vaporization of water at 100 °C is 40.6 kJ mol-1and when 1 mol of water is vaporized at 100 °C and 1 atm pressure, the volume increase is 30.19 dm3. Calculate the work done by the system, the change in internal energy AU, the change in Gibbs' energy AG and the entropy change AS.arrow_forward
- A large electrical power station generates 1000 MW of electricity with an efficiency of 35.0%. (a) Calculate the heat transfer to the power station, Qh , in one day. (b) How much heat transfer Qc occurs to the environment in one day? (c) If the heat transfer in the cooling towers is from 35.0º C water into the local air mass, which increases in temperature from 18.0º C to 20.0º C , what is the total increase in entropy due to this heat transfer? (d) How much energy becomes unavailable to do work because of this increase in entropy, assuming an 18.0º C lowest temperature? (Part of Qccould be utilized to operate heat engines or for simply heating the surroundings, but it rarely is.)arrow_forwardplease answer the following An irreversible process takes place by which the entropy of the universe increases by 1.51 J/K. If the temperature of the environment in which the process occurred is 245 K, how much energy (in J) was made unavailable for useful work as a result?arrow_forward(a) What is the best coefficient of performance for a refrigerator that cools an environment at -28 C and has heat transfer to another environment at 49 ° C? COP, = 3.18 ref (b) How much work must be done for a heat transfer of 4186 kJ from the cold environment? W = 1316.4 kJ (c) What is the cost of doing this if the work costs 10.0 cents per 3.6x10° J (a kilowatt-hour)? Cost in cents = 3.66 (d) How many kJ of heat transfer, Q, occurs into the warm environment? Qn = 1314.5arrow_forward
- Please answer the following question(s): 1. (a) What is the best coefficient of performance for a refrigerator that cools an environment at -28° C and has heat transfer to another environment at 47 ° C? COP ref (b) How much work must be done for a heat transfer of 4186 kJ from the cold environment? W = kj (c) What is the cost of doing this if the work costs 10.0 cents per 3.6 × 106 J (a kilowatt-hour)? Cost in cents = ✓ (d) How many kJ of heat transfer, Qh occurs into the warm environment? Qh= kj Think about what type of refrigerator might operate between these temperatures. Hint: Use the appropriate formula for a refrigerator which is different from a heat pump.arrow_forward100 g of water at 0 C (degrees Celsius) is heated to 100 C. Compute its change in entropy.arrow_forwardOne mole of an ideal gas is heated from 273K to 546K at constant pressure of 1 atmosphere. Compute the work done by the gas in the process.arrow_forward
- An ideal fluid is subjected to a thermodynamic process described by p=CV¯ª and P = np' where p is energy density and P is pressure. For what values of n and I the process is adiabatic if the volume is changed slowly?arrow_forwardThe pV-diagram in Fig. shows a process abc involving0.450 mol of an ideal gas. (a) What was the temperature of this gasat points a, b, and c? (b) How much work was done by or on the gas inthis process? (c) How much heat had to be added during the process toincrease the internal energy of the gas by 15,000 J?arrow_forwardConsider a heat engine that uses reservoirs at 800.°C and 0.00 °C. Calculate the maximum possible efficiency (in %).Answer in 3 Sigfigsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

The Second Law of Thermodynamics: Heat Flow, Entropy, and Microstates; Author: Professor Dave Explains;https://www.youtube.com/watch?v=MrwW4w2nAMc;License: Standard YouTube License, CC-BY