
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20.SE, Problem 19VC
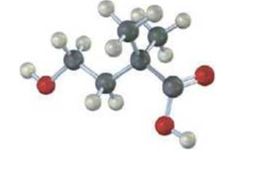
The following
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Laser. Indicate the relationship between metastable state and stimulated emission.
The table includes macrostates characterized by 4 energy levels (&) that are
equally spaced but with different degrees of occupation.
a) Calculate the energy of all the macrostates (in joules). See if they all have
the same energy and number of particles.
b) Calculate the macrostate that is most likely to exist. For this macrostate,
show that the population of the levels is consistent with the Boltzmann
distribution.
macrostate 1 macrostate 2 macrostate 3
ε/k (K) Populations
Populations
Populations
300
5
3
4
200
7
9
8
100
15
17
16
0
33
31
32
DATO: k = 1,38×10-23 J K-1
Don't used Ai solution
Chapter 20 Solutions
Organic Chemistry
Ch. 20.1 - Give IUPAC names for the following compounds:Ch. 20.1 - Draw structures corresponding to the following...Ch. 20.2 - Prob. 3PCh. 20.2 - The Ka for dichloroacetic acid is 3.32 Ă— 10-2....Ch. 20.3 - Calculate the percentages of dissociated and...Ch. 20.4 - Which would you expect to be a stronger acid, the...Ch. 20.4 - Dicarboxylic acids have two dissociation...Ch. 20.4 - The pKa of p-cyclopropylbenzoic acid is 4.45. Is...Ch. 20.4 - Prob. 9PCh. 20.5 - Prob. 10P
Ch. 20.6 - Prob. 11PCh. 20.6 - How might you carry out the following...Ch. 20.7 - Prob. 13PCh. 20.7 - Prob. 14PCh. 20.8 - Cyclopentanecarboxylic acid and...Ch. 20.8 - Prob. 16PCh. 20.SE - Prob. 17VCCh. 20.SE - Prob. 18VCCh. 20.SE - The following carboxylic acid can’t be prepared...Ch. 20.SE - Electrostatic potential maps of anisole and...Ch. 20.SE - Predict the product(s) and provide the mechanism...Ch. 20.SE - Predict the product(s) and provide the mechanism...Ch. 20.SE - Prob. 23MPCh. 20.SE - Predict the product(s) and provide the complete...Ch. 20.SE - Acid-catalyzed hydrolysis of a nitrile to give a...Ch. 20.SE - Prob. 26MPCh. 20.SE - Naturally occurring compounds called cyanogenic...Ch. 20.SE - 2-Bromo-6, 6-dimethylcyclohexanone gives 2,...Ch. 20.SE - Naturally occurring compounds called terpenoids,...Ch. 20.SE - In the Ritter reaction, an alkene reacts with a...Ch. 20.SE - Give IUPAC names for the following compounds:Ch. 20.SE - Prob. 32APCh. 20.SE - Prob. 33APCh. 20.SE - Prob. 34APCh. 20.SE - Prob. 35APCh. 20.SE - Prob. 36APCh. 20.SE - Prob. 37APCh. 20.SE - Prob. 38APCh. 20.SE - Calculate the Ka's for the following acids: (a)...Ch. 20.SE - Thioglycolic acid, HSCH2CO2H, a substance used in...Ch. 20.SE - Prob. 41APCh. 20.SE - Prob. 42APCh. 20.SE - How could you convert butanoic acid into the...Ch. 20.SE - How could you convert each of the following...Ch. 20.SE - How could you convert butanenitrile into the...Ch. 20.SE - How would you prepare the following compounds from...Ch. 20.SE - Prob. 47APCh. 20.SE - Using 13CO2 as your only source of labeled carbon,...Ch. 20.SE - Prob. 49APCh. 20.SE - Which method-Grignard carboxylation or nitrile...Ch. 20.SE - Prob. 51APCh. 20.SE - Prob. 52APCh. 20.SE - Propose a structure for a compound C6H12O2 that...Ch. 20.SE - Prob. 54APCh. 20.SE - How would you use NMR (either 13C or 1H) to...Ch. 20.SE - Prob. 56APCh. 20.SE - A chemist in need of 2,2-dimethylpentanoic acid...Ch. 20.SE - Prob. 58APCh. 20.SE - Prob. 59APCh. 20.SE - Prob. 60APCh. 20.SE - Prob. 61APCh. 20.SE - Prob. 62APCh. 20.SE - Prob. 63APCh. 20.SE - The following pKa values have been measured....Ch. 20.SE - Identify the missing reagents a-f in the following...Ch. 20.SE - Propose a structure for a compound, C4H7N, that...Ch. 20.SE - Prob. 67APCh. 20.SE - The 1H and 13C NMR spectra below belong to a...Ch. 20.SE - Propose structures for carboxylic acids that show...Ch. 20.SE - Carboxylic acids having a second carbonyl group...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In an experiment, the viscosity of water was measured at different temperatures and the table was constructed from the data obtained. a) Calculate the activation energy of viscous flow (kJ/mol). b) Calculate the viscosity at 30°C. T/°C 0 20 40 60 80 η/cpoise 1,972 1,005 0,656 0,469 0,356arrow_forwardDon't used Ai solutionarrow_forwardLet's see if you caught the essentials of the animation. What is the valence value of carbon? a) 4 b) 2 c) 8 d) 6arrow_forward
- A laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).arrow_forwardA laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).arrow_forwardThe number of microstates corresponding to each macrostate is given by N. The dominant macrostate or configuration of a system is the macrostate with the greatest weight W. Are both statements correct?arrow_forward
- For the single step reaction: A + B → 2C + 25 kJ If the activation energy for this reaction is 35.8 kJ, sketch an energy vs. reaction coordinate diagram for this reaction. Be sure to label the following on your diagram: each of the axes, reactant compounds and product compounds, enthalpy of reaction, activation energy of the forward reaction with the correct value, activation energy of the backwards reaction with the correct value and the transition state. In the same sketch you drew, after the addition of a homogeneous catalyst, show how it would change the graph. Label any new line "catalyst" and label any new activation energy.arrow_forwardHow many grams of C are combined with 3.75 ✕ 1023 atoms of H in the compound C5H12?arrow_forwarde. f. CH3O. יון Br NaOCH3 OCH 3 Br H₂Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License