
Chemistry
4th Edition
ISBN: 9780078021527
Author: Julia Burdge
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20.2, Problem 1PPC
Practice Problem  CONCEPTUALIZE
CONCEPTUALIZE
Which of the following graphs best represents the relationship between energy released
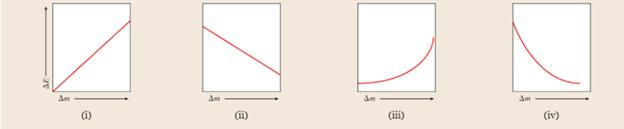
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem 3. Radium-226, which undergoes alpha decay, has a half-life of 1600yr. (a) How many alpha particles are
emitted in 5.0 min by a 10.0-mg sample of 226 Ra ? (b) What is the activity of the sample in mCi ?
Problem 4. Cobalt-60, which undergoes beta decay, has a half-life of 5.26yr. (a) How many beta particles are emitted in
600 s by a 3.75 – mg sample of "Co ? (b) What is the activity of the sample in Bq?
i need clear ans and solve
very very fast in 20 min
and thank you | DYBALA
: A certain container has a 0.56 g of 90Sr. Calculate the activity of the
material in units of curies? (77.3 ci)
Whether or not the process is observed in nature, which of the following could account for the transformation of carbon-10 to boron-10?
(Select all that apply.)
electron capture
alpha decay
positron emission
beta decay
Chapter 20 Solutions
Chemistry
Ch. 20.1 - Practice Problem ATTEMPT
Identify X in each of...Ch. 20.1 - Prob. 1PPBCh. 20.1 - Practice Problem CONCEPTUALIZE
For each process,...Ch. 20.1 - Prob. 1CPCh. 20.1 - Prob. 2CPCh. 20.2 - Prob. 1PPACh. 20.2 - Prob. 1PPBCh. 20.2 - Practice Problem CONCEPTUALIZE Which of the...Ch. 20.2 - Prob. 1CPCh. 20.2 - Prob. 2CP
Ch. 20.2 - Prob. 3CPCh. 20.2 - Prob. 4CPCh. 20.3 - Prob. 1PPACh. 20.3 - Prob. 1PPBCh. 20.3 - Practice Problem CONCEPTUALIZE
The Think About It...Ch. 20.3 - Prob. 1CPCh. 20.3 - Prob. 2CPCh. 20.3 - Prob. 3CPCh. 20.4 - Practice Problem ATTEMPT Determine the age of a...Ch. 20.4 - Practice Problem BUILD How much 206 Pb will be in...Ch. 20.4 - Prob. 1PPCCh. 20.4 - Prob. 1CPCh. 20.4 - Prob. 2CPCh. 20.5 - Prob. 1PPACh. 20.5 - Prob. 1PPBCh. 20.5 - Practice Problem CONCEPTUALIZE
One of the major...Ch. 20 - Prob. 1QPCh. 20 - Prob. 2QPCh. 20 - Prob. 3QPCh. 20 - Prob. 4QPCh. 20 - Prob. 5QPCh. 20 - Prob. 6QPCh. 20 - Prob. 7QPCh. 20 - Prob. 8QPCh. 20 - 20.9 why is it impossible for the isotope to...Ch. 20 - Prob. 10QPCh. 20 - Prob. 11QPCh. 20 - Prob. 12QPCh. 20 - Prob. 13QPCh. 20 - For each pair of isotopes listed, predict which...Ch. 20 - Prob. 15QPCh. 20 - Prob. 16QPCh. 20 - Prob. 17QPCh. 20 - Prob. 18QPCh. 20 - Prob. 19QPCh. 20 - Prob. 20QPCh. 20 - Prob. 21QPCh. 20 - Prob. 22QPCh. 20 - Prob. 23QPCh. 20 - Prob. 24QPCh. 20 - Prob. 25QPCh. 20 - Prob. 26QPCh. 20 - Prob. 27QPCh. 20 - Prob. 28QPCh. 20 - Prob. 29QPCh. 20 - Prob. 30QPCh. 20 - Prob. 31QPCh. 20 - Prob. 32QPCh. 20 - Prob. 33QPCh. 20 - Prob. 34QPCh. 20 - Prob. 35QPCh. 20 - Prob. 36QPCh. 20 - Prob. 37QPCh. 20 - Prob. 38QPCh. 20 - Prob. 39QPCh. 20 - Prob. 40QPCh. 20 - Prob. 41QPCh. 20 - Prob. 42QPCh. 20 - Prob. 43QPCh. 20 - Prob. 44QPCh. 20 - Prob. 45QPCh. 20 - Prob. 46QPCh. 20 - Prob. 47QPCh. 20 - Prob. 48QPCh. 20 - Prob. 49QPCh. 20 - Prob. 50QPCh. 20 - Prob. 51QPCh. 20 - Prob. 52QPCh. 20 - Prob. 53QPCh. 20 - Prob. 54QPCh. 20 - Prob. 55QPCh. 20 - Prob. 56QPCh. 20 - Prob. 57QPCh. 20 - Prob. 58QPCh. 20 - Prob. 59QPCh. 20 - Prob. 60QPCh. 20 - Prob. 61QPCh. 20 - Prob. 62APCh. 20 - Prob. 63APCh. 20 - Prob. 64APCh. 20 - Prob. 65APCh. 20 - Prob. 66APCh. 20 - Prob. 67APCh. 20 - Prob. 68APCh. 20 - Prob. 69APCh. 20 - Prob. 70APCh. 20 - Prob. 71APCh. 20 - Prob. 72APCh. 20 - Prob. 73APCh. 20 - Prob. 74APCh. 20 - Prob. 75APCh. 20 - Prob. 76APCh. 20 - Prob. 77APCh. 20 - Prob. 78APCh. 20 - Prob. 79APCh. 20 - Prob. 80APCh. 20 - Prob. 81APCh. 20 - Prob. 82APCh. 20 - Prob. 83APCh. 20 - Prob. 84APCh. 20 - Prob. 85APCh. 20 - Prob. 86APCh. 20 - Prob. 87APCh. 20 - Prob. 88APCh. 20 - Prob. 89APCh. 20 - Prob. 90APCh. 20 - Prob. 91APCh. 20 - Prob. 92APCh. 20 - Prob. 93APCh. 20 - Prob. 94APCh. 20 - Prob. 95APCh. 20 - Prob. 96APCh. 20 - Prob. 97APCh. 20 - Prob. 98APCh. 20 - Prob. 99APCh. 20 - Prob. 100APCh. 20 - Prob. 101APCh. 20 - Prob. 102APCh. 20 - Prob. 103APCh. 20 - Prob. 1SEPPCh. 20 - Prob. 2SEPPCh. 20 - Prob. 3SEPPCh. 20 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem 1. It takes 5.2 min for a 1.000 - g sample of 210Fr to decay to 0.250 g. What is the half-life of 210Fr? Problem 2. How much time is required for a 6.25 - mg sample of "Cr to decay to 0.75mg if it has a half-life of 27.8 days?arrow_forwardThe ratio between the number of tritium (13T; radio active isotope of hydrogen) and the naturally occurring isotope H is n(T) : n(H) = 1 : 1019 and the half-life of tritium is 12.5 years. Given that the molar mass of water is 18.02 g/mol, how many grams of water is needed to to observe 10 tritium decay events in one hour? Group of answer choices 5.02 g 15.0 g 1.08 g 23.6 g 0.502 g please explainarrow_forwardMY NOTES ASK YOUR TEACHER PRACTICE ANOTHER Consider the following nuclear reaction F -0 + e + v and determine the Q value or total energy released (in MeV) in this process. (Let the atomic mass of 1°F be 18.0009 u, the atomic mass of O be 17.9992 u and the mass of e be 5.4858 x 10-4 u.) Mev Additional Materials O Readingarrow_forward
- Self-Assessment Question If we start with 1.000 g of strontium-90, 0.953 g will remain after 2 years. (a) What is the half-life of strontium-90? (b) how much strontium-90 will remain after 5 years? (c) What is the initial activity of the sample in Bq? THINKING Whee.arrow_forward(11) Lead-211 is a radioactive isotope with a half-life of 36.1 minutes. How long will it take for only 20% of the ( initial amount of lead-211 in a sample to remain? (A) 0.0446 min (B) 0.0192 min asymoonu anton (C) 72.2 min (D) 83.8 min (E) 10.4 minarrow_forwardQuestion 14 How much energy is produced in the creation of 5 grams ofo by the process: 14y - a-H+ !7o (14N= 14.00307 g mole, a=4.0026 g/mol, 'H= 1.00783 g mole, 170=16.99913 g mol)arrow_forward
- Whether or not the process is observed in nature, which of the following could account for the transformation of nitrogen-13 to carbon-13? (Select all that apply.) positron emission alpha decay beta decay electron capturearrow_forward(7) Carbon-14 dating is used in archeology to determine the age of items that were alive or were made from living materials. This works because the proportion of radioactive Carbon-14 to the other isotopes of Carbon is relatively constant in the atmo- sphere. (Volcanos constantly refresh the C-14 content.) A wooden tool from an archeological site is found to contain 22 micrograms of C-14, whereas a similar hnount of modern wood would contain 100 micrograms. How old is the tool? 58 surface weat..pdf JoanneCarbonell .pdf PDFarrow_forwardWhether or not the process is observed in nature, which of the following could account for the transformation of iodine-123 to tellurium-123? (Select all that apply.) positron emission electron capture beta decay alpha decayarrow_forward
- Whether or not the process is observed in nature, which of the following could account for the transformation of chromium-51 to vanadium-51? (Select all that apply.) alpha decay beta decay electron capture positron emissionarrow_forwardQuestion A radioactive waste from a clinical laboratory contains 0.2 µCi (microcuries) of calcium-45 (45Ca) per liter. The half-life of 45Ca is152 days. a. How long must this waste be stored before the level of radioactivity falls below the required maximum of 0.01 µCi/L?arrow_forwardPlease show your solution on a paper using the formulas on the picture. Thank you so much! Question: Fossilized tortoise shell was discovered in an ancient cave dwelling in northern Philippines. A sample of the shell has a specific activity (which is equivalent to Nt) of 4.12 disintegration per minute per gram (d/min-g). If the ratio of C-12 and C-14 in living organisms results to 17.5 d/min-g (which is equivalent to N0), how old is the fossil given that the half-life of C-14 is 5730 years?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY