
Chemistry Atoms First2e
2nd Edition
ISBN: 9781947172647
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20, Problem 24E
Explain, in terms of Figure 20.2, how unstable heavy nuclides (atomic number > 83) may decompose to form nuclides of greater stability (a) if they are below the band of stability and (b) if they are above the band of stability. 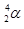
Figure 20.2 This plot shows the nuclides that are known to exist and those are stable. The stable nuclides are indicated in blue, and the unstable nuclides are indicated in green. Note that all
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Chemistry Atoms First2e
Ch. 20 - Write the following isotopes in hyphenated form...Ch. 20 - Write the following isotopes in nuclide notation...Ch. 20 - For the following isotopes that have missing...Ch. 20 - For each of the isotopes in Exercise 20.1,...Ch. 20 - Write the nuclide notation, including charge if...Ch. 20 - Calculate the density of the M1224g nucleus in...Ch. 20 - What are the two principal differences between...Ch. 20 - The mass of the atom N1123a is 22.9893 arm.. (a)...Ch. 20 - Which of the following nuclei lie within the band...Ch. 20 - Which of the following nuclei lie within the band...
Ch. 20 - Write a brief description or definition of each of...Ch. 20 - Which of the various particles ( particles, ...Ch. 20 - Complete each of the following equations by adding...Ch. 20 - Complete each 0f the following equations: (a)...Ch. 20 - Write a balanced equation for each of the...Ch. 20 - Technetium-99 is prepared from 98Mo. Molybdenum-SS...Ch. 20 - The mass of the atom F919 is 18.99840 amu.. (a)...Ch. 20 - For the reaction C614N714+?, if 100.0 g of carbon...Ch. 20 - What are the types of radiation emitted by the...Ch. 20 - That changes occur to the atomic number and mass...Ch. 20 - What is the change in the nucleus that results...Ch. 20 - Many nuclides with atomic numbers greater than 83...Ch. 20 - Why is electron capture accompanied by the...Ch. 20 - Explain, in terms of Figure 20.2, how unstable...Ch. 20 - Which of the following nuclei is most likely to...Ch. 20 - The following nuclei do not lie in the band of...Ch. 20 - The following nuclei do not lie in the band of...Ch. 20 - Predict by what mode(s) of spontaneous radioactive...Ch. 20 - Write a nuclear reaction for each step in the...Ch. 20 - Write a nuclear reaction for each step in the...Ch. 20 - Define the term half-life and illustrate it with...Ch. 20 - A 1.00106 -g sample of nobelium, N102254o, has a...Ch. 20 - 239Pu is a nuclear waste byproduct with a...Ch. 20 - The isotope 208Tl undergoes decay with a...Ch. 20 - If 1.000 g of R88226a produces 0.0001 mL of the...Ch. 20 - The isotope S3890r one of the extremely hazardous...Ch. 20 - Technetium-99 is often used for assessing heart,...Ch. 20 - What is the age of mummified primate skin that...Ch. 20 - A sample of rock was found to contain 8.23 mg of...Ch. 20 - A laboratory investigation shows that a sample of...Ch. 20 - Plutonium was detected in trace amounts in natural...Ch. 20 - A B47e atom (mass = 7.0169 amu) decays into a L37i...Ch. 20 - A B58 atom (mass = 8.0246 amu) decays into B48...Ch. 20 - Isotopes such as 26Al (half-life: 7.2105 years)...Ch. 20 - Write a balanced equation for each of the...Ch. 20 - Write a balanced equation for each of the...Ch. 20 - Write the balanced nuclear equation for the...Ch. 20 - How does nuclear fission differ from nuclear...Ch. 20 - Both fusion and fission are nuclear reactions. Why...Ch. 20 - Cite the conditions necessary for a nuclear chain...Ch. 20 - Describe the components of a nuclear reactor.Ch. 20 - In usual practice, both a moderator and control...Ch. 20 - Describe how the potential energy of uranium is...Ch. 20 - The mass of a hydrogen atom (11H) is 1.007825 arm;...Ch. 20 - How can a radioactive nuclide be used to show that...Ch. 20 - Technetium-99m has a half-life of 6.01 hours. If a...Ch. 20 - Iodine that enters the body is stored in the...Ch. 20 - If a hospital were storing radioisotopes, what is...Ch. 20 - Based on what is known about Radon-222’s primary...Ch. 20 - Given specimens uranium-232 (t1/2=68.9y) and...Ch. 20 - A scientist is studying a 2.234 g sample of...Ch. 20 - Given specimens neon-24 (t1/2=3.38min) and...
Knowledge Booster
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co