
Biology: Science for Life with Physiology (6th Edition) (Belk, Border & Maier, The Biology: Science for Life Series, 5th Edition)
6th Edition
ISBN: 9780134555430
Author: Colleen Belk, Virginia Borden Maier
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 3LTB
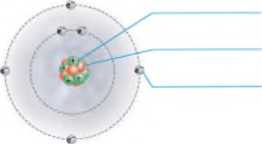
Add labels to the figure that follows, which illustrates the subatomic particles associated with a carbon atom.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Give examples of four different types of bond involving hydrogen atoms. (polar covalent, non-polar covalent, hydrogen bonding and ionic). Comment on any unusual features and consequences of the examples you cite.
#11
The highlighted structure is the?
The configuration of the given structure below is: *
но-сн, о он
но
CH,-OH
OH
alpha-L
alpha-D
beta-L
beta-D
Chapter 2 Solutions
Biology: Science for Life with Physiology (6th Edition) (Belk, Border & Maier, The Biology: Science for Life Series, 5th Edition)
Ch. 2 - Prob. 1LTBCh. 2 - Prob. 2LTBCh. 2 - Add labels to the figure that follows, which...Ch. 2 - Prob. 4LTBCh. 2 - Prob. 5LTBCh. 2 - Which of the following terms is least like the...Ch. 2 - Different proteins are composed of different...Ch. 2 - Proteins may function as ___________ genetic...Ch. 2 - Prob. 9LTBCh. 2 - Eukaryotic cells differ from prokaryotic cells in...
Ch. 2 - Which of the following lists the chemical bonds...Ch. 2 - Which of the following is not consistent with...Ch. 2 - Consider a virus composed of a protein coat...Ch. 2 - Prob. 2AAATBCh. 2 - Carbon, oxygen, hydrogen, and nitrogen are common...Ch. 2 - List some alternate explanations that should be...Ch. 2 - Do some web-based research using scientifically...Ch. 2 - Prob. 1MTC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Below is a diagram illustrating electron shells around a chloride ion. Keeping in mind that chloride has an atomic number of 17, drag the appropriate number of electrons to each electron shell to create a Cl-1 ion. Labels can be used more than once.arrow_forwardDraw a “dot & cross” diagram to show the shape of a water molecule (H2O). Bond angles need to be shown. State the shape of a water molecule.arrow_forwardOrganic molecules all contain carbon. Describe the structure and function of each of these organic molecules. carbohydrates, lipids, proteins, nucleic acids.arrow_forward
- Identify the molecule shown below. You may also see the molecule by clicking on this link. H H HO O phospholipid O DNA ORNA O cholesterol O peptide (protein)arrow_forwardNitrogen is an isotope of nitrogen that has 7 protons and 3 neutrons.What are the atomic atom and atomic mass number of nitrogen 11 here is the picture here.arrow_forwardFill the chart for the 4 Macromolecules.arrow_forward
- fill the following table polymer or large biological molecule monomer or smaller subunit one funtion name od covalent bond nulceic acids three fatty acids easter bond acts as an enzyme immediate or long-term energy source glycosidic linkagearrow_forwardA protein is composed of amino acids, which are held together by the covalent bonds known as: phosphodiester bonds ester bonds glycosidic bonds ether bonds peptide bondsarrow_forwardIdentify the elements that make up the following biomolecules. Write a yes or no in the box to indicate if the element is present or not. BIOMOLECULE carbohydrates proteins lipids carbon hydrogen nitrogen oxygen phosphorus nucleic acidsarrow_forward
- ________ atoms give organic molecules their overall shape; ________ atoms determine the overall chemical behavior of organic molecules.arrow_forwardThe “octet rule” in chemistry helps predict the types of bonds thatatoms will form. In general, an atom will be most stable if it fills itsouter shell of 8 electrons. Atoms with fewer than 4 valence electronstend to donate electrons and those with more than 4 valence electronstend to accept additional electrons; those with exactly 4 can do both.Using this rule, determine what category each of the followingelements falls into: N, S, C, P, O, H, Ca, Fe, and Mg. (You will needto work out the valence of the atoms.)arrow_forward:0-H-1--:N-H Hydrogen Bond Which statement best helps explain the formation of the hydrogen bond represented in the figure? A The oxygen has a partial positive charge, and the nitrogen has a partial negative charge. (B) The nitrogen has a partial negative charge, and the hydrogen attached to the oxygen has a partial positive charge.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license