
Concept explainers
(a)
Interpretation:
The conformation of rings A, B, C and D in Cholestanol has to be described.
Concept Introduction:
The conformation structures of six membered rings are given by chair forms. In the chair conformation, all
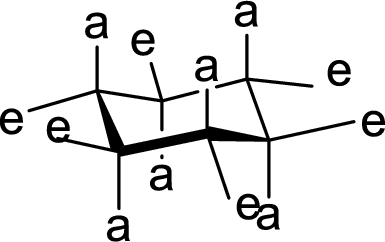
(b)
Interpretation:
The hydroxyl group present on Ring A is either axial or equatorial has to be given.
Concept Introduction:
The
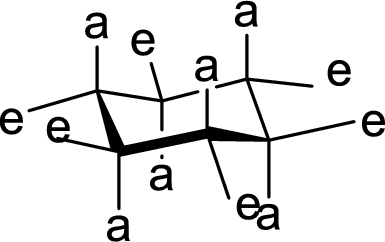
(c)
Interpretation:
The methyl group present at the junction of Ring A and Ring B is either axial or equatorial to Ring A and Ring B has to be given.
Concept Introduction:
The
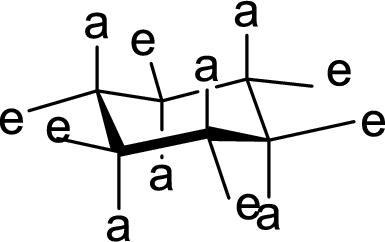
(d)
Interpretation:
The methyl group present at the junction of Ring C and Ring D is either axial or equatorial has to be given.
Concept Introduction:
The
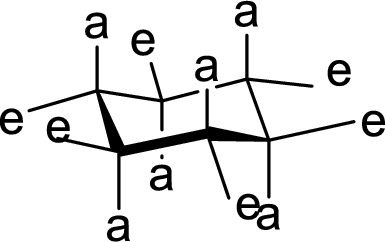
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry
- For each pair of compounds, predict which compound has a higher boiling point. then explain why that compound has a higher boiling point. (a) isopropyl bromide and n-butyl bromide (b) isopropyl chloride and tert-butyl bromide (c) 1-bromobutane and 1-chlorobutanearrow_forward(A) Which of the following ethers is the least soluble in water? (B) Which produces ethanol and methyl iodide after treatment with dilute HI?arrow_forwardCholic acid, a major constituent of bile, has the structure shown.(a) Draw the structure of cholic acid, showing the rings in their chairconformations, and label each methyl group and hydroxy group asaxial or equatorial. (Making a model may be helpful.)(b) Cholic acid is secreted in bile as an amide linked to the aminogroup of glycine. This cholic acid–amino acid combination acts asan emulsifying agent to disperse lipids in the intestines for easierdigestion. Draw the structure of the cholic acid–glycine combination,and explain why it is a good emulsifying agent.arrow_forward
- Describe how would you distinguish the following pairs, (a) Benzene and cyclohexane (b) Phenol and toluene (c) Phenol and benzoic acid (d) methanol and isopropyl alcoholarrow_forwardDraw structures to correspond with the following common and systematic names:(a) phenyl formate (b) cyclohexyl benzoatearrow_forwardBiphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forward
- 1. (a) Describe aromaticity, Kekule structure and resonance structure for benzene. (b) Why is benzene more stable than aliphatic alkenes?arrow_forward(a) Draw the structure of the following :(i) p-Methylbenzaldehyde (ii) 4-Methylpent-3-en-2-one(b) Give chemical tests to distinguish between the following pairs of compounds :(i) Benzoic acid and Ethyl benzoate, (ii) Benzaldehyde and Acetophenone.(iii) Phenol and Benzoic acid.arrow_forwardDescribe how would you distinguish the following pairs, (a) Benzene and cyclohexane (b) Phenol and toluene (c) Phenol and benzoic acidarrow_forward
- Answer ALL parts of this question. (a) Provide the structure and name of the aldehyde with the formula, C2H4O. (b) Draw the two steps of the Strecker synthesis used for the conversion of the aldehyde in part (a) into racemic alanine. (c) Draw the (R) and (S) enantiomers of alanine. Which is the naturally occurring enantiomer? (d) Draw the structure of the dipeptide, Ala-Gly.arrow_forward(a) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 in the presence of Pd-C but does not react with H2 in the presence of Lindlar catalyst. (b) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 when either catalyst is present.arrow_forward6. Describe concisely a chemical test to distinguish between the following pairs of compounds. (a) n-pentanol and 3-methylpentan-3-ol (b) Ethanal dan pentanal (c) Phenol and benzoic acidarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
