
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 14E
Explain why this Lewis structure for CO is not as valid as the Lewis structure you drew in theprevious question even though it has no “hot spots” of + or
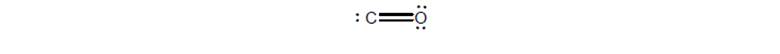
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The carbonate anion, CO32- , is a resonance hybrid. Draw all of the important resonance structures for this molecule. If an atom has a nonzero formal charge, be sure the formal charge is shown clearly in the structure. Use the resonance structures to calculate the average formal charge on each O atom (which are all equivalent in the "true" structure). [Note: all of the important contributing resonance structures have octets around each atom that desires an octet.]
Using only single bonds, draw two corect Lewis structures of HCIO, one with Cl as the
central atom and the other with O as the central atom. Calculate the formal charges of all
atoms in each molecule. State which is the more likely structure based on the formal
charges.
In the previous problem, you drew two structures of HCIO. Are these resonance structures?
Explain why or why not.
Draw the correc (best) Lewis structure (LS) for the ionic compound K2SeO3 (on looose leaf paper--NOT TO BE SUBMITTED) and then answer the following questions.(NOTE: Use the cardinal numbers 0, 1, 2, 3, and so on for any quantity required)a) What charge does the K ion have in this compound? (give size & sign of charge: e.g., 1+, 2-, etc.) b) What charge does the SeO3 ion have in this compound? (give size & sign of charge: e.g., 1+, 2-, etc.) c) How many lines, if any, attach each K ion to the SeO3 ion? d) How many valence electrons does the SeO3 ion (alone) have? e) How many single bonds does the LS of the SeO3 ion have? f) What is the number of lone electron pairs in the LS of the SeO3 ion?
Chapter 2 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 2 - Prob. 1CTQCh. 2 - The valence shell of an atom in a legitimate Lewis...Ch. 2 - Prob. 3CTQCh. 2 - Prob. 4CTQCh. 2 - Prob. 5CTQCh. 2 - It is impossible to draw a legitimate Lewis...Ch. 2 - Describe how to calculate the total number of...Ch. 2 - Prob. 8CTQCh. 2 - Prob. 9CTQCh. 2 - Prob. 10CTQ
Ch. 2 - Prob. 11CTQCh. 2 - Prob. 12CTQCh. 2 - A complete Lewis structure must show all nonzero...Ch. 2 - Prob. 14CTQCh. 2 - Prob. 15CTQCh. 2 - Prob. 16CTQCh. 2 - Prob. 17CTQCh. 2 - Prob. 18CTQCh. 2 - Complete the rest of the table for N, O or X by...Ch. 2 - Prob. 20CTQCh. 2 - Prob. 21CTQCh. 2 - Make a checklist that can be used to determine if...Ch. 2 - Prob. 2ECh. 2 - Prob. 3ECh. 2 - Draw the Lewis structure of a neutral molecule...Ch. 2 - Prob. 5ECh. 2 - For each element, predict (and draw a Lewis...Ch. 2 - Predict which of the following species is least...Ch. 2 - The molecules BH3 and SF6 and the ion SO42 exist...Ch. 2 - These are NOTlegitimate Lewisstructures (and...Ch. 2 - Fill in missing formal charges where needed (all...Ch. 2 - Below each structure in the previous question is a...Ch. 2 - Prob. 12ECh. 2 - Carbon monoxide (CO) is an example of an overall...Ch. 2 - Explain why this Lewis structure for CO is not as...Ch. 2 - Prob. 15ECh. 2 - Prob. 16ECh. 2 - Prob. 17ECh. 2 - Prob. 18ECh. 2 - Prob. 19E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the pyrosulfate ion, S2O72-. It has no sulfur–sulfur nor oxygen–oxygen bonds. (a) Write a Lewis structure for the pyrosulfate ion using only single bonds. (b) What is the formal charge on the sulfur atoms for the Lewis structure you drew in part (a)? (c) Write another Lewis structure using six bonds and two O—S bonds. (d) What is the formal charge on each atom for the structure you drew in part (c)?arrow_forwardCarbon monoxide (CO) is an example of an overall neutral molecule (netcharge=0) that hasnon-zero formal charges. Draw a Lewis structure of carbon monoxide (CO).arrow_forwardWrite all possible resonance structures for the following species. Assign a formal charge to each atom. In each case, which resonance structure is the most important? (a) NO2 (nitrogen is central) (b) ClCNarrow_forward
- Three known isomers exist of N2CO, with the atoms in these sequences: NOCN; ONNC; and ONCN. Write resonance structures for each isomer and use formal charge to predict which isomer is the most stable.arrow_forwardThe electrostatic potential surface for SOCl2 is pictured here. (a) Draw a Lewis electron dot picture for the molecule, and give the formal charge of each atom. (b) What is the molecular geometry of SOCl2? Is it polar?arrow_forwardDraw a Lewis structure for IO4- in which the octet rule is satisfied on all atoms and show all NONZERO formal charges on all atoms. Note that the overall charge on this ion is -1.arrow_forward
- Draw Lewis structures of the four molecules and/or ions with the formulas given below and figure out which two of these are isoelectronic with each another. Species which are isoelectronic have the same number of atoms, the same total number of electrons, the same number of valence electrons, and the same valence electron structure (single and multiple bonds and lone pairs in same places), but need not have the same distribution of formal charges on atoms.CN- and Cl2 and F2 and N2 a) CN- and N2 are isoelectronic.b) F2 and Cl2 are isoelectronic.c) N2 and Cl2 are isoelectronic.d) CN- and Cl2 are isoelectronic.e) N2 and F2 are isoelectronic.arrow_forward+ Write a Lewis structure for the ammonium ion, NH4™. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Include all nonzero formal charges. Do not use the square brackets tool in your answer. P aste opy [*arrow_forwardBased on the atom connectivity shown bellow,evaluate the four resonance structure for the thiosulfate ion S2O3 ^2-. Use curved arrows to indicate how you get from one resonance structure to another. Assign formal changes to all atoms and determine which of these resonance structure is the most stable based on a formal charge analysis Explain your answer thoroughly. Look at the picture.arrow_forward
- Consider the NCO anion. the following three Lewis structures can be drawn for it: Structure 1 Structure 2 Structure 3 [N=C=0] [:N=C_Q] [:N—c=0:] Based on the formal charges determined in the previous three questions, which of these three resonance structures would you predict to be the most stable? Structure 1 Structure 2 Structure 3 Structures 2 and 3 (equally stable) All three structures are equally stable, because all three satisfy the octet rule.arrow_forwardComplete the Lewis structures for COCl₂ and SOCI₂ using the skeletal structure shown below, being sure to follow the procedure for minimizing the sum of the absolute values for the formal charges, where the octet rule need not be followed. Based on the complete structures, which statement below is true? [a_i_d] Cl—N—C Cl-Br Cl Hint: Consider the potential for multiple bonds on the molecules and be sure to account for all valence electrons. O The NOCI₂ exhibits both residual formal charges and resonance hybrids, while the BrOCI₂+ exhibits formal charges but no resonance hybrids. O No answer text provided. The NOC₂ exhibits both formal charges and resonance hybrids, while the BrOCl₂ exhibits resonance hybrids but no formal charges. O At least one atom in each molecule exhibits formal charges, and the molecules have no resonance hybrids. O The BroCl₂ exhibits both formal charges and resonance hybrids, while the POCI₂ exhibits resonance hybrids but no formal charges. O The NOCI₂ exhibits…arrow_forwardDraw the best Lewis structure for the following polyatomic ion and be sure to include all resonance structures and formal charges. Hint: Chlorine is the central atom. chlorate, ClO3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY