
Anatomy & Physiology (6th Edition)
6th Edition
ISBN: 9780134156415
Author: Elaine N. Marieb, Katja N. Hoehn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 12MC
Which of the following covalently bonded molecules are polar?
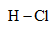
(a)
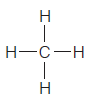
(b)
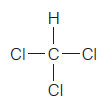
(c)
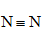
(d)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
B-pt and M-pt of various organic compounds depends on intermolecular forces of attraction which depend on following
(a) Inter molecular/intramolecular H-bond.
(b) Dipole-Dipole interaction (Carboxyl and ether).
(c) Molecular size
(d) Surface area. (branching).
In principle, there are many different, chemically diverse ways in which small molecules can be linked to form polymers. For example, the small molecule ethene (CH2=CH2) is used commercially to make the plastic polyethylene (...–CH2–CH2– CH2–CH2–CH2–...). The individual subunits of the three major classes of biological macromolecules, however, are all linked by similar reaction mechanisms, i.e., by condensation reactions that eliminate water. Can you think of any benefits that this chemistry offers and why it might have been selected in evolution?
Define the following terms:(a) Chiral (b) Achiral(c) Chiral carbon (d) Enantiomer
Chapter 2 Solutions
Anatomy & Physiology (6th Edition)
Ch. 2.1 - What form of energy is found in the food we eat?Ch. 2.1 - What form of energy is used to transmit messages...Ch. 2.1 - What type of energy is available when we are...Ch. 2.2 - What two elements besides H and N make up the bulk...Ch. 2.2 - An element has a mass of 207 and has 125 neutrons...Ch. 2.2 - How do the terms atomic mass and atomic weight...Ch. 2.3 - What is the meaning of the term molecule?Ch. 2.3 - Why is sodium chloride (NaCl) considered a...Ch. 2.3 - Blood contains a liquid component and living...Ch. 2.4 - What kinds of bonds form between water molecules?
Ch. 2.4 - Oxygen (8O) and argon (18A) are both gases. Oxygen...Ch. 2.4 - Assume imaginary compound XY has a polar covalent...Ch. 2.5 - Which reaction type-synthesis, decomposition, or...Ch. 2.5 - Why are many reactions that occur in living...Ch. 2.5 - What specific name is given to decomposition...Ch. 2.6 - Salts are electrolytes. What does that mean?Ch. 2.6 - Which ion is responsible for increased acidity?Ch. 2.6 - To minimize the sharp pH shift that occurs when a...Ch. 2.6 - Prob. 19CYUCh. 2.7 - Prob. 20CYUCh. 2.8 - What are the monomers of carbohydrates called?...Ch. 2.8 - What is the animal form of stored carbohydrate...Ch. 2.9 - Prob. 23CYUCh. 2.10 - What does the name amino acid tell you about the...Ch. 2.10 - What is the primary structure of proteins?Ch. 2.10 - What are the two types of secondary structure in...Ch. 2.10 - How do enzymes reduce the amount of activation...Ch. 2.11 - How do DNA and RNA differ in the bases and sugars...Ch. 2.11 - What are two important roles of DNA?Ch. 2.12 - Glucose is an energy-rich molecule. So why do body...Ch. 2.12 - What change occurs in ATP when it releases energy?Ch. 2 - Which of the following forms of energy is the...Ch. 2 - All of the following are examples of the four...Ch. 2 - The mass number of an atom is (a) equal to the...Ch. 2 - A deficiency in this element can be expected to...Ch. 2 - Which set of terms best describes a proton? (a)...Ch. 2 - The subatomic particles responsible for the...Ch. 2 - Prob. 7MCCh. 2 - Which of the following does not describe a...Ch. 2 - In a beaker of water, the water-water bonds can...Ch. 2 - When a pair of electrons is shared between two...Ch. 2 - Molecules formed when electrons are shared...Ch. 2 - Which of the following covalently bonded molecules...Ch. 2 - Prob. 13MCCh. 2 - Factors that accelerate the rate of chemical...Ch. 2 - Prob. 15MCCh. 2 - Waters importance to living systems reflects (a)...Ch. 2 - Acids (a) release hydroxyl ions when dissolved in...Ch. 2 - Prob. 18MCCh. 2 - Prob. 19MCCh. 2 - A chemical has an amine group and an organic acid...Ch. 2 - Prob. 21MCCh. 2 - Enzymes are organic catalysts that (a) alter the...Ch. 2 - Define or describe energy, and explain the...Ch. 2 - Some energy is lost in energy energy conversion....Ch. 2 - Provide the atomic symbol for each of the...Ch. 2 - Consider the following information about three...Ch. 2 - How many moles of aspirin, C9H8O4, are in a bottle...Ch. 2 - Given the following types of atoms, decide which...Ch. 2 - What are hydrogen bonds and how are they important...Ch. 2 - Prob. 8SAQCh. 2 - Differentiate clearly between primary, secondary,...Ch. 2 - Prob. 10SAQCh. 2 - Describe the mechanism of enzyme action.Ch. 2 - Explain why, if you pour water into a glass very...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Hypoglycin A, an amino acid derivative found in unripened lychee, is an acutely toxic compound that produces seizures, coma, and sometimes death in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?arrow_forward(A) What are Waxes? Draw the structure of wax, which is made up of palmitic acid (16:0) and a saturated 18-carbon alcohol. (В) Consider the structure of menthol. How many isoprene units are present in menthol? (C) What type of isoprene linkage (head-to-tail or tail-to-tail) is present in menthol? Identify the isoprene linkage in the following structure of menthol and indicate by a circle. Menthol =arrow_forwardBradykinin, a peptide that helps to regulate blood pressure, has the primary structure Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg.(a) Draw the complete structural formula of bradykinin.(b) Bradykinin has a very kinked secondary structure.Why?arrow_forward
- Using the appropriate chemical structures describe the monomers and polymers for each of the following macromolecules; a) proteins b) carbohydratesarrow_forwardUsing the appropriate chemical structures describe the monomers and polymers for each of the following macromolecules; a) fats b) nucleic acidarrow_forwardAre the following items chiral or achiral? Give a justification for your answers.(a) A fork (b) This textbook(c) Your right hand(d) A blank 3 X 5 index cardarrow_forward
- (a) What is the net charge at neutral pH of a tripeptide containing only alanine? (b) How does the total number of negative and positive charges change following hydrolysis of the tripeptide?arrow_forwardΦ and ψ in the Ramachandran plot (below) are: a) Rotational angles around the bond between the α-carbon and N-H (Φ) and C=O (ψ). b) Amino acid solubility in octanol (Φ) and water (ψ). c) Hydrogen bond angles in α-helices (Φ) and β-sheets (ψ). d) Amino acid solubility in water (Φ) and octanol (ψ).arrow_forward2) You are studying the tripeptide Lys-Val-Thr. a) Draw the full structure of the tripeptide Lys-Val-Thr at pH 7 and label each of the following in your drawing:i) atoms that are co-planar in one pep de bond ii) one φ (phi) and one ψ (psi) angle b) Could you use the absorbance at 280 nm to determine the concentra on of this tripep de? Briefly explain your answerarrow_forward
- Which of the following is a purine that forms 3 hydrogen bonds with its complementary base? (a) A (b) C (c) G (d) T (e ) Uarrow_forwardWhat form exists at the isoelectric point of each of the following amino acids: (a) valine; (b) leucine; (c) proline; (d) glutamic acid?arrow_forward(b) The structure of a drug called tirbanibulin which is used to treat a pre-cancerous skin condition called actinic keratosis is shown below. (i) (ii) (iii) (iv) ہے مههممنن State Lipinski's Rule of Five (ROF) and Veber's addition to the ROF. Briefly state why the ROF is used in medicinal chemistry?. Given that the molecular mass of tirbanibulin is 431.536 g.mol-¹ and its log P is 3.2, apply the ROF and Veber's additional rule to this drug and show clearly how you arrived at your answer. Comment on the outcome of your analysis of the structure in part (iii).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license