1 Units, Physical Quantities, And Vectors 2 Motion Along A Straight Line 3 Motion In Two Or Three Dimensions 4 Newton’s Laws Of Motion 5 Applying Newton’s Laws 6 Work And Kinetic Energy 7 Potential Energy And Energy Conservation 8 Momentum, Impulse, And Collisions 9 Rotation Of Rigid Bodies 10 Dynamics Of Rotational Motion 11 Equilibrium And Elasticity 12 Fluid Mechanics 13 Gravitation 14 Periodic Motion 15 Mechanical Waves 16 Sound And Hearing 17 Temperature And Heat 18 Thermal Properties Of Matter 19 The First Law Of Thermodynamics 20 The Second Law Of Thermodynamics 21 Electric Charge And Electric Field 22 Gauss’s Law 23 Electric Potential 24 Capacitance And Dielectrics 25 Current, Resistance, And Electromotive Force 26 Direct-current Circuits 27 Magnetic Field And Magnetic Forces 28 Sources Of Magnetic Field 29 Electromagnetic Induction 30 Inductance 31 Alternating Current 32 Electromagnetic Waves 33 The Nature And Propagation Of Light 34 Geometric Optics 35 Interference 36 Diffraction 37 Relativity 38 Photons: Light Waves Behaving As Particles 39 Particles Behaving As Waves 40 Quantum Mechanics I: Wave Functions 41 Quantum Mechanics Ii: Atomic Structure 42 Molecules And Condensed Matter 43 Nuclear Physics 44 Particle Physics And Cosmology expand_more
19.1 Thermodynamic Systems 19.2 Work Done During Volume Changes 19.3 Paths Between Thermodynamic States 19.4 Internal Energy And The First Law Of Thermodynamics 19.5 Kinds Of Thermodynamic Processes 19.6 Internal Energy Of An Ideal Gas 19.7 Heat Capacities Of An Ideal Gas 19.8 Adiabatic Processes For An Ideal Gas Chapter Questions expand_more
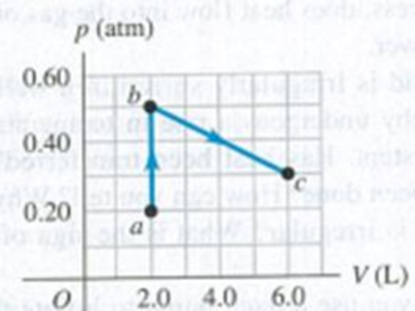
Problem 19.1DQ: For the following processes, is the work done by the system (defined as the expanding or contracting... Problem 19.2DQ Problem 19.3DQ: In which situation must you do more work: inflating a balloon at sea level or inflating the same... Problem 19.4DQ Problem 19.5DQ: Discuss the application of the first law of thermodynamics to a mountaineer who eats food, gets warm... Problem 19.6DQ: When ice melts at 0C, its volume decreases. Is the internal energy change greater than, less than,... Problem 19.7DQ: You hold an inflated balloon over a hot-air vent in your house and watch it slowly expand. You then... Problem 19.8DQ: You bake chocolate chip cookies and put them, still warm, in a container with a loose (not airtight)... Problem 19.9DQ: Imagine a gas made up entirely of negatively charged electrons. Like charges repel, so the electrons... Problem 19.10DQ: In an adiabatic process for an ideal gas, the pressure decreases. In this process does the internal... Problem 19.11DQ: When you blow on the back of your hand with your mouth wide open, your breath feels warm. But if you... Problem 19.12DQ: An ideal gas expands while the pressure is kept constant. During this process, does heat flow into... Problem 19.13DQ: A liquid is irregularly stirred in a well-insulated container and thereby undergoes a rise in... Problem 19.14DQ: When you use a hand pump to inflate the tires of your bicycle, the pump gets warm after a while.... Problem 19.15DQ: In the carburetor of an aircraft or automobile engine, air flows through a relatively small aperture... Problem 19.16DQ: On a sunny day, large bubbles of air form on the sun-warmed earth, gradually expand, and finally... Problem 19.17DQ: The prevailing winds on the Hawaiian island of Kauai blow from the northeast. The winds cool as they... Problem 19.18DQ Problem 19.19DQ: In a constant-volume process, dU = nCV dT. But in a constant-pressure process, it is not true that... Problem 19.20DQ: When a gas surrounded by air is compressed adiabatically, its temperature rises even though there is... Problem 19.21DQ: When a gas expands adiabatically, it does work on its surroundings. But if there is no heat input to... Problem 19.22DQ Problem 19.23DQ: A system is taken from state a to state b along the three paths shown in Fig. Q19.23. (a) Along... Problem 19.24DQ: A thermodynamic system undergoes a cyclic process as shown in Fig. Q19.24. The cycle consists of two... Problem 19.1E: Two moles of an ideal gas are heated at constant pressure from T = 27C to T = 107C. (a) Draw a... Problem 19.2E: Six moles of an ideal gas are in a cylinder fitted at one end with a movable piston. The initial... Problem 19.3E Problem 19.4E: BIO Work Done by the Lungs. The graph in Fig. E19.4 shows a pV-diagram of the air in a human lung... Problem 19.5E: CALC During the time 0.305 mol of an ideal gas undergoes an isothermal compression at 22.0C, 392 J... Problem 19.6E: A gas undergoes two processes. In the first, the volume remains constant at 0.200 m3 and the... Problem 19.7E: Work Done in a Cyclic Process. (a) In Fig. 19.7a, consider the closed loop 1 3 2 4 1. This is a... Problem 19.8E: Figure E19.8 shows a pV-diagram for an ideal gas in which its absolute temperature at b is... Problem 19.9E: A gas in a cylinder expands from a volume of 0.110 m3 to 0.320 m3. Heat flows into the gas just... Problem 19.10E: Five moles of an ideal monatomic gas with an initial temperature of 127C expand and, in the process,... Problem 19.11E: The process abc shown in the pV-diagram in Fig. F.19.11 involves 0.0175 mol of an ideal gas. (a)... Problem 19.12E: A gas in a cylinder is held at a constant pressure of 1.80 105 Pa and is cooled and compressed from... Problem 19.13E: The pV-diagram in Fig. E19.13 shows a process abc involving 0.450 mol of an ideal gas. (a) What was... Problem 19.14E: Boiling Water at High Pressure. When water is boiled at a pressure of 2.00 atm, the heat of... Problem 19.15E: An ideal gas is taken from a to b on the pV-diagram shown in Fig. E19.15. During this process, 700 J... Problem 19.16E: During an isothermal compression of an ideal gas, 410 J of heat must be removed from the gas to... Problem 19.17E: A cylinder contains 0.250 mol of carbon dioxide (CO2) gas at a temperature of 27.0C. The cylinder is... Problem 19.18E: A cylinder contains 0.0100 mol of helium at T = 27.0C. (a) How much heat is needed to raise the... Problem 19.19E: In an experiment to simulate conditions inside an auto-mobile engine. 0.185 mol of air at 780 K and... Problem 19.20E: When a quantity of monatomic ideal gas expands at a constant pressure of 4.00 104 Pa, the volume of... Problem 19.21E: Heat Q flows into a monatomic ideal gas, and the volume increases while the pressure is kept... Problem 19.22E: Three moles of an ideal monatomic gas expands at a constant pressure of 2.50 atm; the volume of the... Problem 19.23E: An experimenter adds 970 J of heat to 1.75 mol of an ideal gas to heat it from 10.0C to 25.0C at... Problem 19.24E: Propane gas (C3Hg) behaves like an ideal gas with = 1.127. Determine the molar heat capacity at... Problem 19.25E: CALC The temperature of 0.150 mol of an ideal gas is held constant at 77.0C while its volume is... Problem 19.26E: Five moles of monatomic ideal gas have initial pressure 2.50 103 Pa and initial volume 2.10 m3... Problem 19.27E: A monatomic ideal gas that is initially at 1.50 103 Pa and has a volume of 0.0800 m3 is compressed... Problem 19.28E: The engine of a Ferrari F355 F1 sports car takes in air at 20.0C and 1.00 atm and compresses it... Problem 19.29E: During an adiabatic expansion the temperature of 0.450 mol of argon (Ar) drops from 66.0C to 10.0C.... Problem 19.30E: A player bounces a basketball on the floor, compressing it to 80.0% of its original volume. The air... Problem 19.31E: On a warm summer day, a large mass of air (atmospheric pressure 1.01 105 Pa) is heated by the... Problem 19.32E: A cylinder contains 0.100 mol of an ideal monatomic gas, Initially the gas is at 1.00 105 Pa and... Problem 19.33P: A quantity of air is taken from state a to state b along a path that is a straight line in the... Problem 19.34P: One-half mole of an ideal gas is taken from state a to state c as shown in Fig. P19.34. (a)... Problem 19.35P: Figure P19.35 shows the pV-diagram for a process in which the temperature of the ideal gas remains... Problem 19.36P: The graph in Fig. P19.36 shows a pV-diagram for 3.25 mol of ideal helium (He) gas. Part ca of this... Problem 19.37P: When a system is taken from state a to state b in Fig. P19.37 along path acb, 90.0 J of heat flows... Problem 19.38P: A thermodynamic system is taken from state a to state c in Fig. P19.38 along either path abc or path... Problem 19.39P: A volume of air (assumed to be an ideal gas) is first cooled without changing its volume and then... Problem 19.40P: Three moles of argon gas (assumed to be an ideal gas) originally at 1.50 104 Pa and a volume of... Problem 19.41P: Two moles of an ideal monatomic gas go through the cycle abc. For the complete cycle, 800 J of heat... Problem 19.42P: Three moles of an ideal gas are taken around cycle acb shown in Fig. P19.42. For this gas, Cp = 29.1... Problem 19.43P: Figure P19.43 shows a pV-diagram for 0.0040 mol of ideal H2 gas. The temperature of the gas does not... Problem 19.44P: (a) Onc-third of a mole of He gas is taken along the path abc shown in Fig. P19.44. Assume that the... Problem 19.45P: Starting with 2.50 mol of N2 gas (assumed to be ideal) in a cylinder at 1.00 atm and 20.0C, a... Problem 19.46P: Nitrogen gas in an expandable container is cooled from 50.0C to 10.0C with the pressure held... Problem 19.47P: CALC A cylinder with a frictionless, movable piston like that shown in Fig. 19.5 contains a quantity... Problem 19.48P: CP A Thermodynamic Process in a Solid. A cube of copper 2.00 cm on a side is suspended by a string.... Problem 19.49P Problem 19.50P: High-Altitude Research. A large research balloon containing 2.00 103 m3 of helium gas at 1.00 atm... Problem 19.51P: An air pump has a cylinder 0.250 m long with a movable piston. The pump is used to compress air from... Problem 19.52P: A certain ideal gas has molar heat capacity at constant volume CV. A sample of this gas initially... Problem 19.53P: A monatomic ideal gas expands slowly to twice its original volume, doing 450 J of work in the... Problem 19.54P: CALC A cylinder with a piston contains 0.250 mol of oxygen at 2.40 105 Pa and 355 K. The oxygen may... Problem 19.55P: Use the conditions and processes of Problem 19.54 to compute (a) the work done by the gas, the heat... Problem 19.56P: CALC A cylinder with a piston contains 0.150 mol of nitrogen at 1.80 105 Pa and 300 K. The nitrogen... Problem 19.57P: Use the conditions and processes of Problem 19.56 to compute (a) the work done by the gas, the heat... Problem 19.58P: Comparing Thermodynamic Processes. In a cylinder, l.20 mol of an ideal monatomic gas. initially at... Problem 19.59P: DATA You have recorded measurements of the heat flow Q into 0.300 mol of a gas that starts at T1 =... Problem 19.60P: DATA You compress a gas in an insulated cylinderno heat flows into or out of the gas. The gas... Problem 19.61P: DATA You place a quantity of gas into a metal cylinder that has a movable piston at one end. No gas... Problem 19.62CP Problem 19.63PP: BIO ANESTHETIC GASES. One type of gas mixture used in anesthesiology is a 50%/50% mixture (by... Problem 19.64PP: BIO ANESTHETIC GASES. One type of gas mixture used in anesthesiology is a 50%/50% mixture (by... Problem 19.65PP: BIO ANESTHETIC GASES. One type of gas mixture used in anesthesiology is a 50%/50% mixture (by... Problem 19.66PP: BIO ANESTHETIC GASES. One type of gas mixture used in anesthesiology is a 50%/50% mixture (by... format_list_bulleted



 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning



