
Concept explainers
What alcohol can be oxidized to each
a. 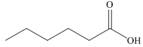 b.
b. 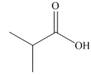 c.
c. 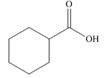
(a)
Interpretation: Alcohol that can be oxidized to the given carboxylic acid is to be identified.
Concept introduction: Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. They are polar in nature due to electronegativity difference between the atoms in a compound. They sometimes exist as a dimer. Dimers are the compounds that consist of two monomer units connected by bonds or forces. Carboxylic acids can be synthesized from alkynes, alkene, benzene derivatives, alcohol and allylic halides by using different reagents.
Answer to Problem 19.10P
Alcohol that can be oxidized to the given carboxylic acid is
Explanation of Solution
The structure of alcohol that can be oxidized to the given carboxylic acid is shown below.
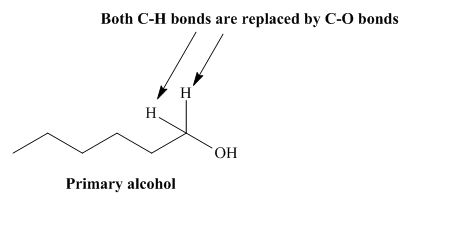
Figure 1
The compound in the figure is
Alcohol that can be oxidized to the given carboxylic acid is
(b)
Interpretation: Alcohol that can be oxidized to the given carboxylic acid is to be identified.
Concept introduction: Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. They are polar in nature due to electronegativity difference between the atoms in a compound. These sometimes exist as a dimer. Dimers are the compounds that consist of two monomer units connected by bonds or forces. Carboxylic acids can be synthesized from alkynes, alkene, benzene derivatives, alcohol and allylic halides by using different reagents.
Answer to Problem 19.10P
Alcohol that can be oxidized to the given carboxylic acid is
Explanation of Solution
The given carboxylic acid is
Alcohol that can be oxidized to the given carboxylic acid is
(c)
Interpretation: Alcohol that can be oxidized to the given carboxylic acid is to be identified.
Concept introduction: Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. They are polar in nature due to electronegativity difference between the atoms in a compound. These sometimes exist as a dimer. Dimers are the compounds that consist of two monomer units connected by bonds or forces. Carboxylic acids can be synthesized from alkynes, alkene, benzene derivatives, alcohol and allylic halides by using different reagents.
Answer to Problem 19.10P
Alcohol that can be oxidized to the given carboxylic acid is
Explanation of Solution
The structure of alcohol that can be oxidized to the given carboxylic acid is shown below.
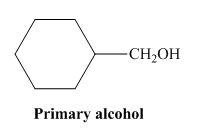
Figure 2
The compound in the figure is
Alcohol that can be oxidized to the given carboxylic acid is
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry
- 18. Identify the lactone formed by the following hydroxy carboxylic acid. A. A B. B C. C D. D OH OH & & A) B) C) D)arrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forward17. Which functional groups are in the following molecule of aspirin? HO, a benzene ring, an ester, a ketone, and an alcohol b. a benzene ring, two ketones, an ether, and an alcohol a benzene ring, a carboxylic acid, an ether, and a ketone d. a benzene ring, a carboxylic acid, and an ester a. c.arrow_forward
- part: 6b: a,b,carrow_forwardare acetals and ketals stable to acid or base? Why?arrow_forwardAn acid catalyst in nucleophilic addition of aldehydes and ketones is used for: Select one: a. Protonation of carbonyl carbon b. Making the aldehyde and ketone more susceptible to nucleophiles c. Increasing the nucleophilicity of the nucleophile d. To provide a medium for the reaction.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





