
Concept explainers
(a)
Interpretation:
The products formed when given compound is treated with
Concept introduction:
Benzene undergoes electrophile substitution. The kinetics of the electrophilic substitution reaction depends upon the nature of substituent present on the benzene ring. Electron releasing groups activates the ring towards the electrophilic substitution reaction while electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
Answer to Problem 18.15P
The product formed by the reaction of given compound with
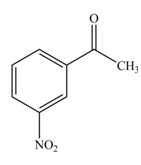
The reaction occurs slower because benzene ring contains deactivating group.
Explanation of Solution
Electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
The substituent present in the given compound is electron withdrawing group. Thus, it directs the electrophile to meta position and deactivates the ring towards the electrophilic substitution reaction. Hence, the given compound reacts slower than benzene. The reaction is shown below.
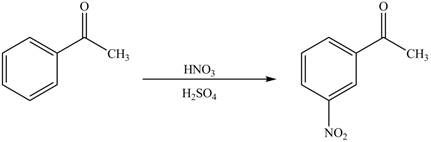
Figure 1
The product formed by the reaction of given compound with
(b)
Interpretation:
The products formed when given compound is treated with
Concept introduction:
Benzene undergoes electrophile substitution. The kinetics of the electrophilic substitution reaction depends upon the nature of substituent present on the benzene ring. Electron releasing groups activates the ring towards the electrophilic substitution reaction while electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
Answer to Problem 18.15P
The product formed by the reaction of given compound with
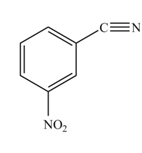
The reaction occurs slower because benzene ring contains deactivating group.
Explanation of Solution
Electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
The substituent present in the given compound is electron withdrawing group. Thus it directs the electrophile to meta position and deactivates the ring towards the electrophilic substitution reaction. Hence, the given compound reacts slower than benzene. The reaction is shown below.
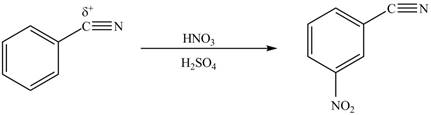
Figure 2
The product formed by the reaction of given compound with
(c)
Interpretation:
The products formed when given compound is treated with
Concept introduction:
Benzene undergoes electrophile substitution. The kinetics of the electrophile substitution reaction depends upon the nature of substituent present on the benzene ring. Electron releasing groups activates the ring towards the electrophilic substitution reaction while electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
Answer to Problem 18.15P
The product formed by the reaction of given compound with
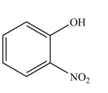
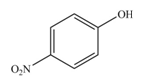
The reaction occurs faster because benzene ring contains activating group.
Explanation of Solution
Electron releasing group directs the electrophile to ortho and para position.
The substituent present in the given compound is electron donating group. Thus, it directs the electrophile to ortho and para position and activates the ring towards the electrophilic substitution reaction. Hence, the given compound reacts faster than benzene. The reaction is shown below.
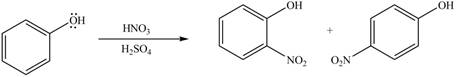
Figure 3
The product formed by the reaction of given compound with
(d)
Interpretation:
The products formed when given compound is treated with
Concept introduction:
Benzene undergoes electrophile substitution. The kinetics of the electrophilic substitution reaction depends upon the nature of substituent present on the benzene ring. Electron releasing groups activates the ring towards the electrophilic substitution reaction while electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
Answer to Problem 18.15P
The products formed by the reaction of given compound with
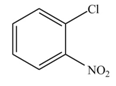
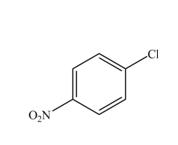
The reaction occurs slower benzene ring because benzene ring contains deactivating group.
Explanation of Solution
The substituent present in the given compound is
Among these two cases, mesomeric effect predominates over inductive effect. Hence, chlorine on benzene ring acts as releasing group but deactivates the benzene ring due to its
Thus, it directs the electrophile to ortho and para position and deactivates the ring towards the electrophilic substitution reaction. Hence, the given compound reacts slower than benzene. The reaction is shown below.
Figure 4
The product formed by the reaction of given compound with
(e)
Interpretation:
The products formed when given compound is treated with
Concept introduction:
Benzene undergoes electrophile substitution. The kinetics of the electrophile substitution reaction depends upon the nature of substituent present on the benzene ring. Electron releasing groups activates the ring towards the electrophilic substitution reaction while electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.
Answer to Problem 18.15P
The products formed by the reaction of given compound with
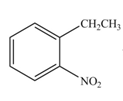
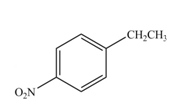
The reaction occurs higher because benzene ring contains activating group.
Explanation of Solution
Electron releasing group directs the electrophile to ortho and para positions.
The substituent present in the given compound is electron withdrawing group. Thus it directs the electrophile to ortho and para positions and activates the ring towards the electrophilic substitution reaction. Hence, the given compound reacts faster than benzene. The reaction is shown below.
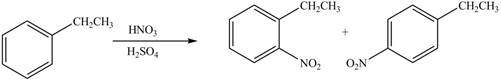
Figure 5
The product formed by the reaction of given compound with
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry
- Hydration of aldehydes and ketones can be catalyzed by acid or base. Bases catalyze hydration by: protonating the carbonyl oxygen making the carbonyl group more electrophilic employing hydroxide ion, which is a better nucleophile than water making the carbonyl group less electrophilic shifting the equilibrium position of the reaction to favor productsarrow_forward1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-octen-3-ol into a less effective molecule. Draw the structure of a hydrogenation product of 1-octen-3-ol.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardwhich reagents complete the reaction?arrow_forward
- What ester is formed when each carboxylic acid is treated with ethanol (CH 3CH 2OH) in the presence of H 2SO 4?arrow_forwardDraw the product of the reaction between a ketone and an alcohol. Include all hydrogen atoms in the product. How would you classify the product of the reaction? Note that a hemiacetal formed from a ketone is also called a hemiketal; an acetal formed from a ketone is also called a ketal. The product is an alcohol. The product is a ketal The product is a ketone. The product is a hemiketal.arrow_forwardAddition of hydrogen (H2) to an aldehyde produces a(an)arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

