
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
13th Edition
ISBN: 9780134421353
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.5, Problem 16.45PP
The following graph shows the activity versus pH curves for pepsin, sucrose, and trypsin. Estimate the optimum pH for each.
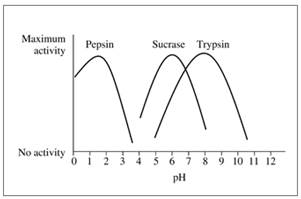
16.46 Refer to the graph in problem 16.45 to determine if the reaction rate in each condition will be at the optimum rate or not.
- Trypsin, pH 5.0
- Sucrase, pH 5.0
- Pepsin, pH 4.0
- Trypsin, pH 8.0
- Pepsin, pH 2.0
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A process that occurs in the human body is shown in the diagram (arrow's direction points to
the direction of this hydrolysis reaction). What would happen if a temperature change
-caused the shape of the enzyme's active site to be altered?
+
Active
Site
Dipeptide
(Substrate)
)-(
Enzyme
Enzyme-substrate Amino
complex
acids
Dipeptide
Breakdown
) - G
The dipeptide would hydrolyze slower or not at all
O The amino acids would combine slower or not at all
The dipeptide would hydrolyze (break down) faster
There will be no effect on the rate of this hydrolysis reaction
The amino acids would combine faster
+
}
Enzyme
Enzymes that are rather aggressive, and turned "off" by a protein
segment until they are activated.
е.
the conversion of the amino acid phenylalanine into non-harmful tyrosine is mutated and
doesn't work as well (or at all). Phenylalanine will still break down into tyrosine by itself,
ol because this is a favorable reaction. Why is the non-functional enzyme a problem? Explain in a
few sentences.
3.
The disease Phenylketonuria (or PKU) occurs when the enzyme that helps to catalyze
O p HOo (pn) O pe oHe
avl lls sew.i Snde (a)H noitas on ol woisd sidst nos stolgno ta
T b e sd linw ps fertt sro.nalom ni encbeono mundiups sis novi 2noitetnono
uTeeg os s oulev
THI
utinogmet
cseo.o
8880
4.
Consider the following reaction, which is exothermic:
C2H4 (g) + Br2 (g) → C2H¾Br2 (g)
ΔΗ<
Predict the effect of each of the following perturbations (shift right/products, shift left/reactants, or
no effect) using Le Chatelier's principle
a.
The reaction temperature is increased:
b. The reaction volume is decreased:
C.
More bromine gas is added:
d. The reaction is done in the presence of helium:
e. C2H4BR2…
Chapter 16 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Ch. 16.1 - Classify each of the following proteins according...Ch. 16.1 - Prob. 16.2PPCh. 16.1 - Prob. 16.3PPCh. 16.1 - Prob. 16.4PPCh. 16.1 - Draw the structure for each of the following amino...Ch. 16.1 - Draw the structure for each of the following amino...Ch. 16.1 - Draw the strcture for each of the following amino...Ch. 16.1 - Prob. 16.8PPCh. 16.1 - Prob. 16.9PPCh. 16.1 - Prob. 16.10PP
Ch. 16.2 - Prob. 16.11PPCh. 16.2 - Prob. 16.12PPCh. 16.2 - Prob. 16.13PPCh. 16.2 - Prob. 16.14PPCh. 16.2 - Prob. 16.15PPCh. 16.2 - Prob. 16.16PPCh. 16.3 - Prob. 16.17PPCh. 16.3 - Prob. 16.18PPCh. 16.3 - Prob. 16.19PPCh. 16.3 - Prob. 16.20PPCh. 16.3 - What type of interaction would you expect between...Ch. 16.3 - What type of interaction would you expect between...Ch. 16.3 - Prob. 16.23PPCh. 16.3 - Prob. 16.24PPCh. 16.3 - Prob. 16.25PPCh. 16.3 - Prob. 16.26PPCh. 16.3 - Prob. 16.27PPCh. 16.3 - Indicate the changes in secondary and tertiary...Ch. 16.4 - Why do chemical reactions in the body require...Ch. 16.4 - Prob. 16.30PPCh. 16.4 - Prob. 16.31PPCh. 16.4 - Prob. 16.32PPCh. 16.4 - Prob. 16.33PPCh. 16.4 - Prob. 16.34PPCh. 16.4 - Prob. 16.35PPCh. 16.4 - 16.36 Match the terms (1) active site, (2)...Ch. 16.4 - Prob. 16.37PPCh. 16.4 - Prob. 16.38PPCh. 16.4 - Prob. 16.39PPCh. 16.4 - Prob. 16.40PPCh. 16.4 - For problems 16.39 to 16.42, see Chemistry Link to...Ch. 16.4 - Prob. 16.42PPCh. 16.5 - Trypsin, a peptidase that hydrolyzes polypeptides,...Ch. 16.5 - pepsin, a peptidase that hydrolyzes proteins,...Ch. 16.5 - The following graph shows the activity versus pH...Ch. 16.5 - The following graph shows the activity versus pH...Ch. 16.5 - Prob. 16.47PPCh. 16.5 - Prob. 16.48PPCh. 16.5 - Prob. 16.49PPCh. 16.5 - Prob. 16.50PPCh. 16.5 - What is the chemical formula for hydroxyurea?Ch. 16.5 - What is the molar mass of hydroxyurea?Ch. 16.5 - Prob. 16.53PPCh. 16.5 - Prob. 16.54PPCh. 16 - Prob. 16.55UTCCh. 16 - Prob. 16.56UTCCh. 16 - Prob. 16.57UTCCh. 16 - Prob. 16.58UTCCh. 16 - 16.59 Identify the amino acids and type of...Ch. 16 - What type of interaction would you expect between...Ch. 16 - Draw the condensed structural formula for...Ch. 16 - Draw the condensed structural formula for...Ch. 16 - Seed and vegetables are often deficient in one or...Ch. 16 - 16.64 Seeds and vegetables are often deficient in...Ch. 16 - Prob. 16.65APPCh. 16 - Prob. 16.66APPCh. 16 - Prob. 16.67APPCh. 16 - Prob. 16.68APPCh. 16 - Prob. 16.69APPCh. 16 - Why do enzymes function only under mild...Ch. 16 - Prob. 16.71APPCh. 16 - Prob. 16.72APPCh. 16 - Prob. 16.73APPCh. 16 - Prob. 16.74APPCh. 16 - Prob. 16.75APPCh. 16 - Prob. 16.76APPCh. 16 - Prob. 16.77APPCh. 16 - Prob. 16.78APPCh. 16 - If a blood test indicates a high level of LDH and...Ch. 16 - Prob. 16.80APPCh. 16 - Prob. 16.81CPCh. 16 - Prob. 16.82CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The human body has an average pH of about 7 and a temperature of about 37C. Use graphs to illustrate enzyme activity in the human body as a function of the following: a. Substrate concentration b. Enzyme concentration c. pH include pH optimum value d. Temperature include temperature optimum valuearrow_forwardsac ve ha inhihi- The following graph shows the activity versus pH curves for pepsin, sucrase, and trypsin. Estimate the optimum pH for each. Maximum activity No activity Pepsin Sucrase Trypsin 01234 567 pH 8 9 10 11 12 16.46 Refer to the graph in problem 16.45 to determine if the reaction rate in each condition will be at the optimum rate or not. a. trypsin, pH 5.0 d. trypsin, pH 8.0 b. sucrase, pH 5.0 c. pepsin, pH 4.0 e. pepsin, pH 2.0arrow_forwardChymotrypsin is secreted by the pancreas and passed into the intestine. The optimal pH for this enzyme is 7.8. If a patient’s pancreas cannot manufacture chymotrypsin, would it be possible to supply it orally? Justifyarrow_forward
- Substrate Enzyme-substrate complex 1. Is this an example of a lock & key or induced fit model of an enzyme? > Enzymearrow_forwardNote the Myoglobin O₂ binding curve below. YO2 1.00 0.80 0.60 0.40 0.20 0.0 Myoglobin O₂ Binding Curve 0 2 5 1.6 moles of O₂ 1.0 moles of O₂ 0.4 moles of O₂ 0.2 moles of O₂ 10 15 20 25 30 po₂ (torr) How much O₂ is released by 4 moles of myoglobin moving from pO₂ = 10 torr to pO₂ = 2 torr?arrow_forwardFind an example of an enzyme. a) What is the chemical reaction that is catalyzed by the enzyme? b) Why is this enzyme/chemical reaction important to the organisms in which it occurs?arrow_forward
- Which of the following enzyme functional groups is in a form in which it could serve as a general acid catalyst to enhance a reaction rate? B. A. H₂C OH B C O OE -NH2 C. H₂C H-NN-H D. H₂C ·0™ E. α-NH₂arrow_forwardThe above diagram illustrates ___? The relationship between the proteome and the metabolome The relationship between the concentration of enzymes and the rate of chemical reactions they catalyze a and barrow_forward13. A specific enzyme will usually only catalyze one particular reacton. True Falsearrow_forward
- Explain the distinction between a general acid catalyst and a specific acid catalyst. Explain how an acid or base provided by an amino acid side chain can catalyze a reaction.arrow_forward3. Some medications permanently inactivate their target enzyme by forming a covalent bond to an amino acid in the enzyme's active site. Aspirin is one example of a pharmaceutical that uses this type of mechanism. So is penicillin. As you saw in the last chapter, the active portion of penicillin is the B-lactam ring. Penicillin forms a covalent bond to serine in the active site transpeptidase, an enzyme that is essential for forming the bacterial cell wall. Propose a mechanism for the inactivation of transpeptidase by penicillin and show the final product of the reaction. NH OH CH3 CH2 CH3 backbone penicillin serinearrow_forwardJuan performed the same chemical reaction twice. His data from the first at- tempt is indicated by the red line. His data from the second attempt is indicated by the blue line. Energy During a Chemical Reaction Energy A B reactants e.g. C6H12O6 + O₂ C without enzyme Juan added pressure. with enzyme Reaction coordinate What change did Juan most likely make to his reaction the second time he per- formed it? activation energy with enzyme D Juan added a catalyst. Juan increased the surface area of the reactants. Juan cooled his reactants. activation energy without enzyme overall energy released during reaction products CO,+H,Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY