
Interpretation:
The reason of faster reaction of magnesium with 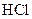 than iron needs to be explained.
than iron needs to be explained.
Concept introduction:
Activity series of metals is the list of metals arranged according to their reactivity. The highly reactive metals are placed at the top of the list and the least reactive metals are placed at the bottom. Potassium is highest reactive metal and gold is the least reactive metal.
Answer to Problem 13SSC
Reactivity of magnesium is more than iron. So, it undergoes reaction with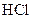 faster.
faster.
Explanation of Solution
The reaction rate depends upon the reactivity. Highly reactive metals undergo reaction faster than the least reactive metals. Magnesium and iron both are metals. Magnesium is placed after potassium in the activity series and the iron is placed lower than magnesium. So, magnesium is more reactive than iron and thus, it undergoes reaction with 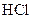 at faster rate than iron.
at faster rate than iron.
Chapter 16 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: The Central Science (14th Edition)
CHEMISTRY-TEXT
Chemistry: The Central Science (13th Edition)
Organic Chemistry (9th Edition)
Organic Chemistry
Chemistry: A Molecular Approach (4th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





