
Interpretation:
The time required to produce 12 mL of oxygen gas at 288K needs to be determined.
Concept introduction:
Rate of a
Answer to Problem 59A
Time required to yield 12 mL of oxygen at 288K is
Explanation of Solution
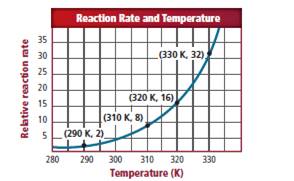
Consider the graph of the relative
The given data points from the graph is as follows: (290K, 2), (310K, 8), (320K, 16) and (330K, 32). The temperature is raised by 10K. Therefore, it is clear that the reaction rate should double from 2 to 4.
At temperature 298K,
Since At temperature 298K 12 mL oxygen gas is produced. Therefore,
At 308 K,
Since the reaction rate is doubled for every 10K increases in temperature.
Consider the reactant rate at 288 K. At a temperature of 298K, to produce 12 mL oxygen it takes 100 s. Since the reaction rate is halved for a decrease in temperature of 10K. Therefore, at 288K it takes twice as long to produce 12 mL of oxygen.
Therefore, the time required to yield 12 mL of oxygen at 288K is
The time required to yield 12 mL of oxygen at 288K is
Chapter 16 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: A Molecular Approach
CHEMISTRY-TEXT
Organic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
General, Organic, and Biological Chemistry (3rd Edition)
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





