
(a)
Interpretation:
The differences in the structural formula of testosterone and methandrostenolone should be determined along with
Concept Introduction:
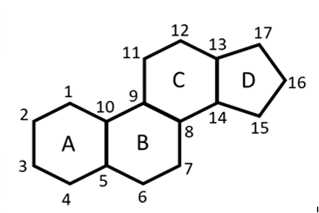
Steroids are the third major class of lipids. These are the compounds containing the following ring system:
Answer to Problem 70P
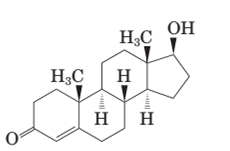
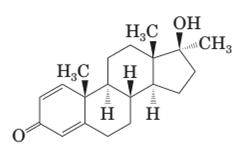
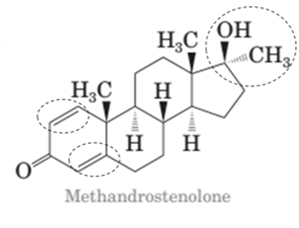
Both methandrostenolone and testosterone hormone contains hydroxyl functional group (alcohol) and carbonyl functional group (kentone).
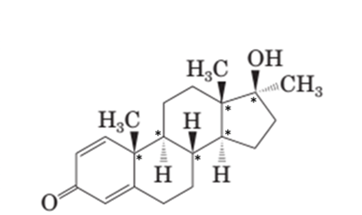
In the structural formula of testosterone hormone, there is only one double bond and it has only one methyl group attached to cyclopentane ring whereas in methandrostenolone there are two double bonds and two methyl groups are attached to the cyclopentane ring.
Explanation of Solution
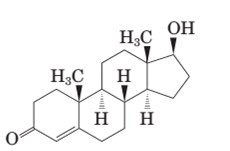
Testosterone and methandrostenolone both the hormones contain one −OH group attached to cyclopentane ring and one >C=O group in cyclohexene ring. Therefore both alcoholic and
In the structure of testosterone there is only a double bond in cyclohexene ring having ketonic group and no methyl group is present on the carbon of cyclopentane ring having alcoholic group as shown:
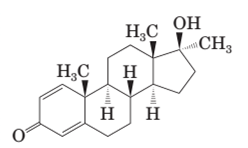
In the structure of methandrostenolone, there are two double bonds in cyclohexene ring having ketonic group and one methyl group attached on the carbon of cyclopentane ring having alcoholic group as shown:
(b)
Interpretation:
In both the given hormones mark all the stereocenters and state the number of stereoisomer possible for each.
Concept Introduction:
Stereoisomers are the compounds that are differ only in the spatial arrangement of their atoms. Each stereoisomer has at least one stereocenter. A stereocenter is a tetrahedral carbon atom that has four different groups bonded to it. For a molecule with n sterocenters, a maximum of
Answer to Problem 70P
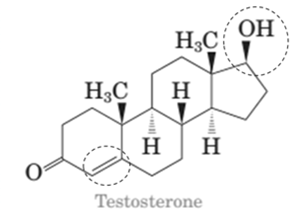
Stereocenters in testosterone:
Since testosterone has six stereocenters therefore it has 64 possible stereoisomers.
Stereocenters in methandrostenolone:
Since methandrostenolone has six stereocenters therefore it has 64 possible stereoisomers.
Explanation of Solution
A stereocenter is a tetrahedral carbon atom that has four different groups bonded to it.
Stereocenters in testosterone:
In testosterone there are six stereocenters.
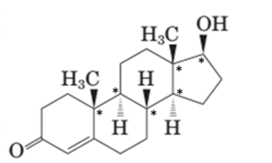
For a molecule with n stereocenter, a maximum of
Stereocenters in methandrostenolone:
In methandrostenolone there are six stereocenters.
For a molecule with n stereocenter, a maximum of
Want to see more full solutions like this?
Chapter 16 Solutions
Introduction To General, Organic, And Biochemistry
- Indicate the correct answer.a) In boranes, the B-B bonds are the most reactive.b) The B-H-B bonds are the reactive centers in the B2H6 molecule.arrow_forwardIn boranes, the B-B bonds are the most reactive.arrow_forwardThe B-H-B bonds are the reactive centers in the B2H6 molecule. Correct?arrow_forward
- Please help I only have two hours left!arrow_forwardBriefly explain inverse spinels.arrow_forwardCalculate the solubility at 25 °C of AgBr in pure water and in 0.34 M NaCN. You'll probably find some useful data in the ALEKS Data resource. Round your answer to 2 significant digits. Solubility in pure water: Solubility in 0.34 M NaCN: 7.31 × 10 M x10 Омarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

