
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN: 9781133104261
Author: Raymond A. Serway, John W. Jewett
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 56P
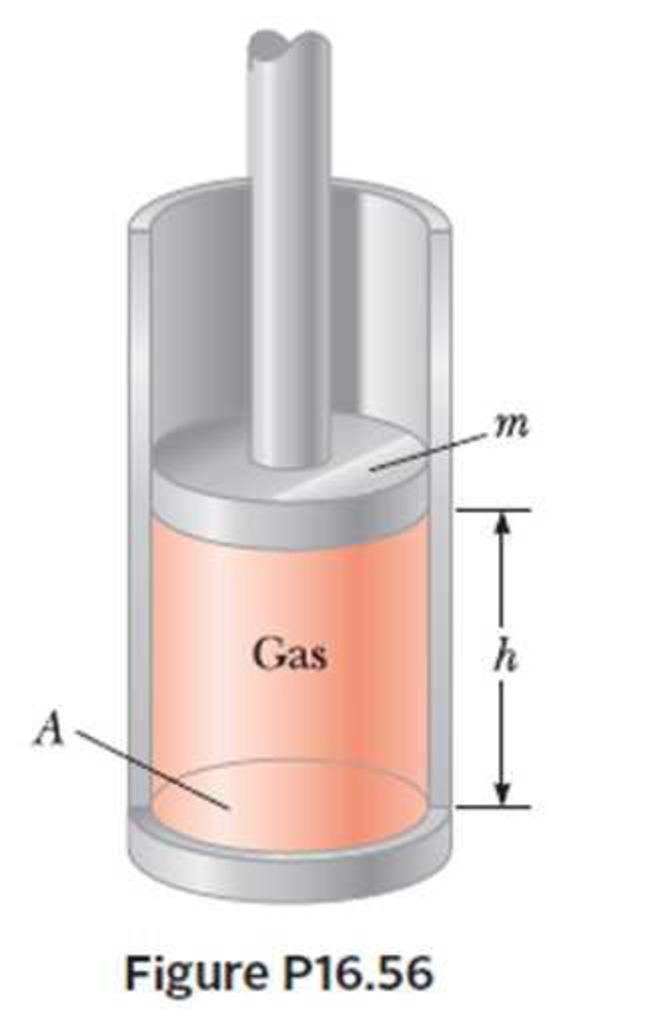
A vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P16.56). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find die height h in Figure P16.56. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
**64. A gas fills the right portion of a
horizontal cylinder whose radius is
5.00 cm. The initial pressure of the gas
- Unstrained
spring
Pin
is 1.01 × 10$ Pa. A frictionless mov-
able piston separates the gas from the
left portion of the cylinder, which is
evacuated and contains an ideal spring, as the drawing shows. The piston
is initially held in place by a pin. The spring is initially unstrained, and
the length of the gas-filled portion is 20.0 cm. When the pin is removed
and the gas is allowed to expand, the length of the gas-filled chamber
doubles. The initial and final temperatures are equal. Determine the
spring constant of the spring.
A gas fills the right portion of a horizontal cylinder whose
radius is 5.80 cm. The initial pressure of the gas is 2.64 x 105
Pa. A frictionless movable piston separates the gas from the
left portion of the cylinder that is evacuated and contains an
ideal spring, as the drawing shows. The piston is initially held in
place by a pin. The spring is initially unstrained, and the length
of the gas-filled portion is 27.1 cm. When the pin is removed
and the gas is allowed to expand, the length of the gas-filled
chamber doubles. The initial and final temperatures are equal.
Determine the spring constant of the spring.
Number i
-Unstrained
spring Pin
Units N/m
A hand-driven tire pump has a piston with a 2.00 cm diameter and a maximum stroke of 25.0 cm.
a. How much work (in J) do you do in one stroke if the average gauge pressure is 2.40 ✕ 105 N/m2 (about 35 psi)?
b.
What average force (in N) do you exert on the piston, neglecting friction and gravity?
Chapter 16 Solutions
Principles of Physics: A Calculus-Based Text
Ch. 16.2 - Consider the following pairs of materials. Which...Ch. 16.3 - Prob. 16.2QQCh. 16.4 - A common material for cushioning objects in...Ch. 16.4 - Prob. 16.4QQCh. 16.5 - Two containers hold an ideal gas at the same...Ch. 16.6 - Prob. 16.6QQCh. 16 - Prob. 1OQCh. 16 - A cylinder with a piston holds 0.50 m3 of oxygen...Ch. 16 - A hole is drilled in a metal plate. When the metal...Ch. 16 - When a certain gas under a pressure of 5.00 106...
Ch. 16 - Prob. 5OQCh. 16 - Prob. 6OQCh. 16 - What would happen if the glass of a thermometer...Ch. 16 - Prob. 8OQCh. 16 - A gas is at 200 K. If we wish to double the rms...Ch. 16 - Prob. 10OQCh. 16 - Prob. 11OQCh. 16 - A rubber balloon is filled with 1 L of air at 1...Ch. 16 - Prob. 13OQCh. 16 - An ideal gas is contained in a vessel at 300 K....Ch. 16 - Prob. 15OQCh. 16 - Prob. 16OQCh. 16 - Prob. 17OQCh. 16 - A sample of gas with a thermometer immersed in the...Ch. 16 - Prob. 19OQCh. 16 - Prob. 1CQCh. 16 - Prob. 2CQCh. 16 - Prob. 3CQCh. 16 - A piece of copper is dropped into a beaker of...Ch. 16 - Prob. 5CQCh. 16 - Prob. 6CQCh. 16 - Prob. 7CQCh. 16 - Prob. 8CQCh. 16 - Prob. 9CQCh. 16 - Prob. 10CQCh. 16 - Prob. 11CQCh. 16 - Prob. 12CQCh. 16 - Prob. 13CQCh. 16 - Prob. 1PCh. 16 - Convert the following to equivalent temperatures...Ch. 16 - Prob. 3PCh. 16 - Prob. 4PCh. 16 - Prob. 5PCh. 16 - Prob. 6PCh. 16 - Prob. 7PCh. 16 - Prob. 8PCh. 16 - Prob. 9PCh. 16 - A sample of a solid substance has a mass m and a...Ch. 16 - Each year thousands of children are badly burned...Ch. 16 - Prob. 12PCh. 16 - Prob. 13PCh. 16 - Prob. 14PCh. 16 - The active element of a certain laser is made of a...Ch. 16 - Prob. 16PCh. 16 - Prob. 17PCh. 16 - Prob. 18PCh. 16 - Prob. 19PCh. 16 - Prob. 20PCh. 16 - Prob. 21PCh. 16 - Prob. 22PCh. 16 - Prob. 23PCh. 16 - Prob. 24PCh. 16 - Prob. 25PCh. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Prob. 28PCh. 16 - The mass of a hot-air balloon and its cargo (not...Ch. 16 - Prob. 30PCh. 16 - A popular brand of cola contains 6.50 g of carbon...Ch. 16 - Prob. 32PCh. 16 - At 25.0 m below the surface of the sea, where the...Ch. 16 - To measure how far below the ocean surface a bird...Ch. 16 - Prob. 35PCh. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - A cylinder contains a mixture of helium and argon...Ch. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - (a) How many atoms of helium gas fill a spherical...Ch. 16 - Fifteen identical particles have various speeds:...Ch. 16 - From the MaxwellBoltzmann speed distribution, show...Ch. 16 - Prob. 47PCh. 16 - Helium gas is in thermal equilibrium with liquid...Ch. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - A mercury thermometer is constructed as shown in...Ch. 16 - A liquid with a coefficient of volume expansion ...Ch. 16 - A clock with a brass pendulum has a period of...Ch. 16 - A vertical cylinder of cross-sectional area A is...Ch. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - The rectangular plate shown in Figure P16.60 has...Ch. 16 - In a chemical processing plant, a reaction chamber...Ch. 16 - Prob. 62PCh. 16 - Prob. 63PCh. 16 - Two concrete spans that form a bridge of length L...Ch. 16 - A 1.00-km steel railroad rail is fastened securely...Ch. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - Consider an object with any one of the shapes...Ch. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - A cylinder that has a 40.0-cm radius and is 50.0...Ch. 16 - Prob. 75P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P18.40). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find the height h in Figure P18.40. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder. Figure P18.40arrow_forwardA gas is in a container of volume V0 at pressure P0. It is being pumped out of the container by a piston pump. Each stroke of the piston removes a volume Vs through valve A and then pushes the air out through valve B as shown in Figure P19.74. Derive an expression that relates the pressure Pn of the remaining gas to the number of strokes n that have been applied to the container. FIGURE P19.74arrow_forwardThe mass of a single hydrogen molecule is approximately 3.32 1027 kg. There are 5.64 1023 hydrogen molecules in a box with square walls of area 49.0 cm2. If the rms speed of the molecules is 2.72 103 m/s, calculate the pressure exerted by the gas.arrow_forward
- A hand—driven tire pump has a piston with a 2.50cm diameter and a maximum stroke of 30.0 cm. (a) How much work do you do in one stroke if the average gauge pressure is 2.40105N/m2 (about 35 psi)? (b) What average force do you exert on the piston, neglecting friction and gravitational force?arrow_forwardA cylinder that has a 40.0-cm radius and is 50.0 cm deep is filled with air at 20.0C and 1.00 atm (Fig. P10.74a). A 20.0-kg piston is now lowered into the cylinder, compressing the air trapped inside as it takes equilibrium height hi (Fig. P16.74b). Finally, a 25.0-kg dog stands on the piston, further compressing the air, which remains at 20C (Fig. P16.74c). (a) How far down (h) does the piston move when the dog steps onto it? (b) To what temperature should the gas be warmed to raise the piston and dog back to hi?arrow_forwardA car engine moves a piston with a circular cross-section of 73000.002cm in diameter a distance of 3.2500.001cm to compress the gas in the cylinder. (a) By what amount is the gas decreased in volume in cubic centimeters? (b) Find the uncertainty in this volume.arrow_forward
- A hand-driven tire pump has a piston with a 2.50-cm diameter and a maximum stroke of 30.0 cm. (a) How much work do you do in one stroke if the average gauge pressure is 2.1105 N/m2 (about 35 psi)? (b) What average force do you exert on the piston, neglecting friction and gravitational force?arrow_forward(a) Given that air is 21% oxygen, find the minimum atmospheric pressure that gives a relatively safe partial pressure of oxygen of 0.16 atm. (b) What is the minimum pressure that gives a partial pressure of oxygen above the quickly fatal level of 0.06 atm? (c) The air pressure at the summit of Mount Everest (8848 m) is 0.334 atm. Why have a few people climbed it without oxygen, while some who have tried, even though they had trained at high elevation, had to tum back?arrow_forwardAn ideal gas is trapped inside a tube of uniform cross-sectional area sealed at one end as shown in Figure P19.49. A column of mercury separates the gas from the outside. The tube can be turned in a vertical plane. In Figure P19.49A, the column of air in the tube has length L1, whereas in Figure P19.49B, the column of air has length L2. Find an expression (in terms of the parameters given) for the length L3 of the column of air in Figure P19.49C, when the tube is inclined at an angle with respect to the vertical. FIGURE P19.49arrow_forward
- A vertical cylindrical tank contains 1.80 mol of an ideal gas under a pressure of 0.300 atm at 20.0 C. The round part of the tank has a radius of 10.0 cm, and the gas is supporting a piston that can move up and down in the cylinder without friction. There is a vacuum above the piston. (a) What is the mass of this piston? (b) How tall is the column of gas that is supporting the piston?arrow_forwardA fluid at a pressure of 3 bar and with a specific volume of 0.18 m°/kg contained in a cylinder behind a piston expands reversibly to a pressure of 0.6 bar according to the law p =2 v² where C is a constant. Calculate the work done by the fluid on the piston.arrow_forwardA hand-driven tire pump has a piston with a 3.00 cm diameter and a maximum stroke of 35.0 cm. (a) How much work (in J) do you do in one stroke if the average gauge pressure is 2.40 x 105 N/m2 (about 35 psi)? (b) What average force (in N) do you exert on the piston, neglecting friction and gravity? Narrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning


Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College
Thermal Expansion and Contraction of Solids, Liquids and Gases; Author: Knowledge Platform;https://www.youtube.com/watch?v=9UtfegG4DU8;License: Standard YouTube License, CC-BY