
Concept explainers
Write chemical equations, showing all necessary reagents, for the preparation of
by each of the following methods:
Hydroboration–oxidation of an
Use of a Grignard reagent
Use of a Grignard reagent in a way different from part (b)
Reduction of a
Hydrogenation of an
Reduction with sodium borohydride
Interpretation:
The chemical equations showing all necessary reagents for the preparation of
Concept introduction:
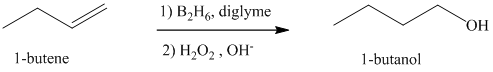
Hydroboration-oxidation of an alkene results in overall addition of water molecule across the double bond with a regioselectivity opposite to that of Markovnikov’s rule.
In the overall reaction, a hydrogen atom gets attached to the double bonded carbon atom having fewer hydrogens and the hydroxyl group gets attached to the carbon atom having greater number of hydrogens.
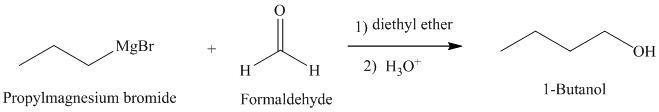
When a Grignard reagent reacts with formaldehyde primary alcohols are produced.
Grignard reagents are nucleophilic and react with oxiranes producing alcohols.
Aldehydes are reduced to the corresponding primary alcohols by using
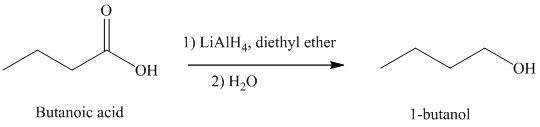
By a suitable reducing agent like lithium aluminum hydride, carboxylic acids are reduced to the corresponding primary alcohols.
Answer to Problem 16P
Solution:
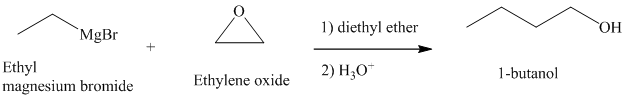

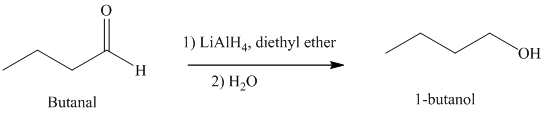
Explanation of Solution
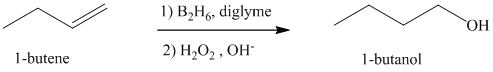
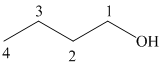
a) Hydroboration-oxidation of an alkene to form
The structure of
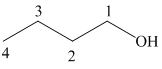
The hydroxyl group is attached to the C1 carbon atom. This alcohol is produced by hydroboration-oxidaton of an alkene. So in the alkene, the C1 carbon atom must be double bonded to the the C2 carbon atom. Thus, the alkene must be
The regioselectivity of hydroboration-oxidation is such that the hydrogen atom will get attached to the C2 carbon atom while the hydroxyl group will get attached to the C1 carbon atom producing
The reaction is shown below:
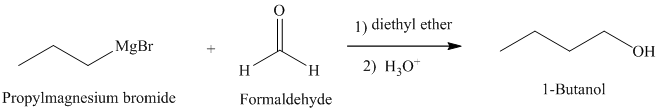
b) Grignard reagent is used to prepare
The structure of
Grignard reagents are nucleophilic and react with carbonyl groups forming a new carbon-carbon bond. An aqueous acid is used to convert the intermediate alkoxy ion to the corresponding alcohol.
In
The reaction is shown below:
c) Different Grignard reagent is used to prepare
The structure of
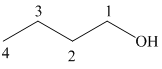
It is a primary alcohol. The hydroxyl group is attached to the C1 carbon atom.
Grignard reagents reacts with ethylene oxide to produce primary alcohol containing two more carbon atoms than the alkyl halide.
Thus, ethyl magnesium bromide, upon reaction with ethylene oxide, will produce
The reaction is shown below:
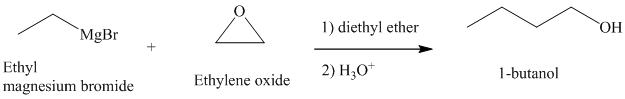
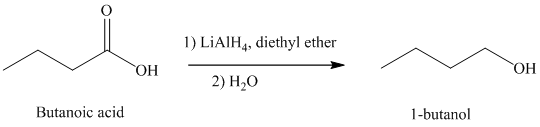
d) The reduction of carboxylic acid to produce

Carboxylic acids, upon reduction, produce the correspionding primary alcohols.
Thus, reduction of butanoic acid with Lithium aluminum hydride will produce
The reaction is shown below:
e) Hydrogenation of an aldehyde to form

Reduction of aldehydes with a suitable reducing agent produces the corresponding primary alcohols.
Thus, reduction of butanal in the presence of Lithium aluminum hydride will produce
The reaction is shown below:
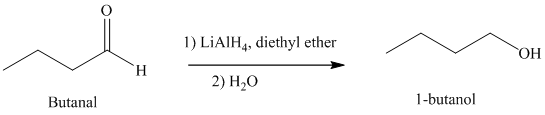
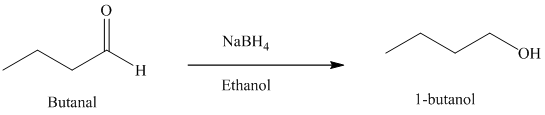
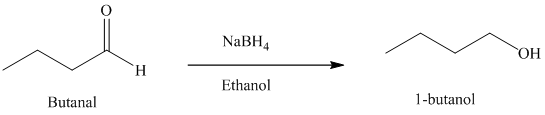
f) Reduction of an aldehyde to form

Sodium borohydride is used for the reduction of aldehydes and ketones to primary and secondary alcohols respectively.
Thus, reduction of butanal with sodium borohydride will produce
The reaction is shown below:
Want to see more full solutions like this?
Chapter 16 Solutions
Solutions Manual for Organic Chemistry
- Aldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forward17-60 1-Propanol can be prepared by the reduction of an aldehyde, but it cannot be prepared by the acid catalyzed hydration of an alkene. Explain why it cannot be prepared from an alkene.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forward
- Show how to synthesize carboxylic acids from oxidation of alcohols and aldehydes,carboxylation of Grignard reagents, oxidative cleavage of alkenes and alkynes,hydrolysis of nitriles, and oxidation of alkylbenzenesarrow_forwardWrite the chemical equation showing reactants, products and catalysts needed (if any) for thefollowing reactions. Write the IUPAC name of the product right beside the structure. a) Reaction of two phenol b) Reaction of 2-bromophenol with sodium hydroxide c) Reaction of potassium phenoxide with 2-chloropentane d) Reaction of isopropyl propyl ether with HBr e) Reaction of 3-methylpentan-3-ol with sulfuric acid f) Dehydration of 1,5-hepta-diolg) Oxidation of 1-decanolh) Hydration od hepta-2,3-dienearrow_forwardEsterification of Carboxylic Acids Provide the reactions (chemical equations) on esterification of the following acids: Ethanoic acid Butanoic acid Benzoic acid Acetic acid + methanol Acetic acid + ethanol Salicylic acid + methanol Acetic acid + benzyl alcoholarrow_forward
- Acid-catalyzed dehydration of 3-methyl-2-pentanol gives three alkenes: 3-methyl-1-pentene, 3-methyl-2-pentene, and 3-methylenepentane. Draw the structure of the carbocation intermediate leading to the formation of 3-methyl-2-pentene.arrow_forwardWrite in the correct reagents for these transformations: a) cyclohexanol -→ chlorocyclohexane b) 2-butanone 2-butanol ward 2-butanol 2-butanone toluene - → benzoic acid nitrobenzene → anilinearrow_forwardDetermine the structure of the methyl ester and organomagnesium bromide reagents that can be combined to form the following alcohol: Part 1: Draw the bond-line formula for the methyl ester reagent. Disregard stereochemistry. Part 2: Draw the bond-line formula for the organomagnesium bromide reagent. Disregard stereochemistry.arrow_forward
- Describe a sequence of reactions by which cis-2-pentene could be prepared from acetylene.arrow_forwardA synthetic organic molecule, G, which contains both aldehyde and ether functional groups, is subjected to a series of reactions in a multi-step synthesis pathway. In the first step, G undergoes a Wittig reaction, leading to the formation of an alkene, H. Subsequently, H is treated with an ozone (O3) reagent followed by a reducing agent in an ozonolysis reaction, resulting in the formation of two different products, I and J. Considering the functional groups present in G and the nature of the reactions involved, what are the most probable structures or functional groups present in products I and J? A. I contains a carboxylic acid group, and J contains an aldehyde group. B. I contains a ketone group, and J contains an alcohol group. C. I and J both contain aldehyde groups. D. I contains an ester group, and J contains a ketone group. Don't use chat gpt.arrow_forwardExplains the general concept of the manufacture of benzene by dealkylation of toluene with hydrogen.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


