
Concept explainers
Interpretation:
The formation of a precipitate when the solution containing 
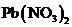 is mixed with
is mixed with 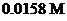
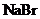 to form a solution is to be predicted.
to form a solution is to be predicted.
Concept introduction: The molar solubility represents the number of moles of a substance which can be dissolved in a liter of solution. The molar solubility is calculated by the solubility product constant 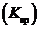
The reaction quotient 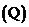 for the reaction is the one through which an ionic compound dissolves is the product of the concentrations of the ionic components raised to their
for the reaction is the one through which an ionic compound dissolves is the product of the concentrations of the ionic components raised to their
If 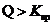 then the solution is supersaturated and the solution forms precipitate under many circumstances.
then the solution is supersaturated and the solution forms precipitate under many circumstances.
To predict: The formation of a precipitate when the solution containing 
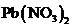 is mixed with
is mixed with 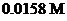
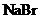 to form a solution.
to form a solution.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Principles of Chemistry: A Molecular Approach (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





