
Chemistry
4th Edition
ISBN: 9780078021527
Author: Julia Burdge
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.10, Problem 1PPC
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
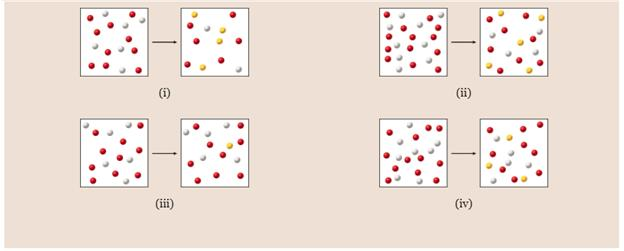
Each of the following diagrams shows a system before and after equilibrium is established. Indicate which of the diagrams best represents a system in which you can neglect the x in the solution, as you did in Sample Problem 15.10.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
CHEMWORK
The reaction H,(g) + I(g) 2 2HI(g) has K, = 45.9 at 763 K. A particular equilibrium mixture at 763 K contains HI at a pressure of 3.28 atm and H, at a pressure of 1.20 atm. Calculate the equilibrium pressure of I,(g) in this mixture.
atm
PL
Submit
Submit Answer
Try Another Version
1 item attempt remaining
(Previous
ywww.yahoo.com 2h A
Top Story
New research raises doubts about 1 st
COVID-19 death count
Cengage Learning | Cengage Technical Support
DELL
The reaction H,(g)+ I(g) 2HI(g) has K, = 45.9 at 763 K. A particular equilibrium mixture at 763 K contains HI at a pressure of 1.10 atm and H2 at a pressure of 3.29 atm. Calculate the equilibrium pressure of I,(g) in this mixture.
atm
P1,
PI.
Submit
Try Another Version
1 item attempt remaining
Submit Answer
pr
1 pt
1 pr
1 pt
Previous
Nex
Email Instructor
Save a
Cengage Learning Cengage Technical Support
国
DELL
Consult the table below and answer the questions that follow.
The table below shows the equilibrium % of product
temperature of 10°C and a pressure of 10bar. A, B and C are all gass
temperature
Pressure (bar)
B) X will be smaller than
40
C) Y will be larger than 40
D) Y will be smaller than 40
b) If the reaction of is represented by
answers is best:
A) X will be larger than 40
B) X will be smaller than
40
C) Y will be larger than
D) Y will be smaller than
40
10
200
40
C in a mixture of A, B and C
es at room
a) If the reaction is EXOTHERMIC, which of the following answe
A) X will be larger than
40
Temperature
10
40
100
X
swers is best:
A+ 1/2B ==> 2C which of the following
at a
Chapter 15 Solutions
Chemistry
Ch. 15.1 - Practice Problem ATTEMPT
In an analysis of the...Ch. 15.1 - Prob. 1PPBCh. 15.1 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.2 - Practice ProblemATTEMPT Write the reaction...Ch. 15.2 - Practice Problem BUILD
Write the equation for the...Ch. 15.2 - Practice ProblemCONCEPTUALIZE In principle, in the...Ch. 15.2 - Select the correct equilibrium expression for the...Ch. 15.2 - Prob. 2CPCh. 15.3 - Practice Problem ATTEMPT Write equilibrium...Ch. 15.3 - Practice Problem BUILD
Which of the following...
Ch. 15.3 - Prob. 1PPCCh. 15.3 - Prob. 1CPCh. 15.3 - Prob. 2CPCh. 15.3 - Given the following information: HF ( a q ) ⇄ H +...Ch. 15.3 - Prob. 4CPCh. 15.4 - Practice ProblemATTEMPT The following reactions...Ch. 15.4 - Practice Problem BUILD
The equation represents a...Ch. 15.4 - Practice ProblemCONCEPTUALIZE Consider a chemical...Ch. 15.4 - Use the following information to answer questions...Ch. 15.4 - Prob. 2CPCh. 15.4 - 15.4.3 If for the reaction at a certain...Ch. 15.4 - If K c = 3 for the reaction X + 2Y ⇄ Z at a...Ch. 15.5 - Practice ProblemATTEMPT Write K? expressions for (...Ch. 15.5 - Prob. 1PPBCh. 15.5 - Prob. 1PPCCh. 15.5 - Prob. 1CPCh. 15.5 - Prob. 2CPCh. 15.5 - Prob. 3CPCh. 15.5 - Prob. 4CPCh. 15.5 - Prob. 5CPCh. 15.5 - Prob. 6CPCh. 15.6 - Practice Problem ATTEMPT
For the reaction:
....Ch. 15.6 - Practice ProblemBUILD K p = 2.79 × 10 − 5 for the...Ch. 15.6 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.7 - Prob. 1PPACh. 15.7 - Prob. 1PPBCh. 15.7 - Prob. 1PPCCh. 15.8 - Practice ProblemATTEMPT Calculate the equilibrium...Ch. 15.8 - Practice ProblemBUILD Determine the initial...Ch. 15.8 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.9 - Prob. 1PPACh. 15.9 - Prob. 1PPBCh. 15.9 - Prob. 1PPCCh. 15.10 - Practice ProblemATTEMPT Aqueous hydrocyanic acid...Ch. 15.10 - Practice Problem BUILD Consider a weak acid, HA,...Ch. 15.10 - Practice ProblemCONCEPTUALIZE Each of the...Ch. 15.11 - Practice Problem ATTEMPT Determine the equilibrium...Ch. 15.11 - Prob. 1PPBCh. 15.11 - Prob. 1PPCCh. 15.12 - Practice ProblemATTEMPT For each change indicated,...Ch. 15.12 - Prob. 1PPBCh. 15.12 - Practice ProblemCONCEPTUALIZE Consider the...Ch. 15.13 - Practice Problem ATTEMPT
For each reaction,...Ch. 15.13 - Practice Problem BUILD
For the following...Ch. 15.13 - Practice Problem CONCEPTUALIZE
Consider the...Ch. 15.14 - Practice Problem ATTEMPT
The reaction of carbon...Ch. 15.14 - Practice Problem BUILD
Consider the hypothetical...Ch. 15.14 - Practice Problem CONCEPTUALIZE
The decomposition...Ch. 15 - The K a for hydrocyanic acid ( HCN ) is 4 .9 × 10...Ch. 15 - 15.2
Determine the concentrations of in a...Ch. 15 - 15.3
Determine the for a weak acid if a 0.10-M...Ch. 15 - Prob. 4KSPCh. 15 - Define equilibrium. Give two examples of a dynamic...Ch. 15 - 15.2 Which of the following statements is correct...Ch. 15 - 15.3 Consider the reversible reaction Explain how...Ch. 15 - What is the law of mass action?Ch. 15 - Briefly describe the importance of equilibrium in...Ch. 15 - Define reaction quotient. How does it differ from...Ch. 15 - Write reaction quotients for the following...Ch. 15 - Write the equation for the reaction that...Ch. 15 - Consider the reaction: 2NO ( g ) + 2H 2 ( g ) ⇄ N...Ch. 15 - The equilibrium constant for the reaction: 2SO 2 (...Ch. 15 - Consider the following equilibrium process at...Ch. 15 - The equilibrium constant for the reaction: 2 H 2 (...Ch. 15 - The first diagram represents a system at...Ch. 15 - These two diagrams represent systems at...Ch. 15 - Define homogeneous equilibrium and heterogeneous...Ch. 15 - What do the symbols K c and K p represent?Ch. 15 - Write the expressions for the equilibrium...Ch. 15 - Write equilibrium constant expressions for K c ,...Ch. 15 - Write the equilibrium constant expressions for K c...Ch. 15 - 15.20 Write the equation relating to , and define...Ch. 15 - 15.21 The equilibrium constant () for the...Ch. 15 - What is K p at 1273°C for the reaction 2CO ( g ) +...Ch. 15 - 15.23 The equilibrium constant for the...Ch. 15 - 15.24 Consider the reaction:
If the equilibrium...Ch. 15 - 15.25 A reaction vessel contains at equilibrium...Ch. 15 - 15.26 The equilibrium constant Kc for the...Ch. 15 - At equilibrium, the pressure of the reacting...Ch. 15 - The equilibrium constant K p for the reaction: PCl...Ch. 15 - Ammonium carbamate ( NH 4 CO 2 NH 2 ) decomposes...Ch. 15 - The following equilibrium constants were...Ch. 15 - 15.31 At a certain temperature, the following...Ch. 15 - 15.32 Pure phosgene gas , was placed in a 1.50-L...Ch. 15 - Consider the equilibrium: 2 NOBr( g ) ⇄ 2 NO( g...Ch. 15 - The following equilibrium constants have been...Ch. 15 - 15.35 The following equilibrium constants have...Ch. 15 - 15.36 The equilibrium constant for the reaction at...Ch. 15 - The following diagrams represent the equilibrium...Ch. 15 - 15.38 Outline the steps for calculating the...Ch. 15 - 15.39 The equilibrium constant K? for the...Ch. 15 - 15.40 For the synthesis of ammonia:
the...Ch. 15 - For the reaction: H 2 ( g ) + CO 2 ( g ) ⇄ H 2 O (...Ch. 15 - At 1000 K, a sample of pure NO, gas decomposes:...Ch. 15 - The equilibrium constant K c for the reaction H 2...Ch. 15 - The dissociation of molecular iodine into iodine...Ch. 15 - The equilibrium constant Kc for the decomposition...Ch. 15 - 15.46 Consider the following equilibrium process...Ch. 15 - 15.47 Consider the heterogeneous equilibrium...Ch. 15 - The equilibrium constant K c for the reaction: H 2...Ch. 15 - The aqueous reaction: L-glutamate + pyruvate ⇄...Ch. 15 - 15.50 Explain Le Châtelier’s principle. How does...Ch. 15 - Use Le Chatelier's principle to explain why the...Ch. 15 - 15.52 List four factors that can shift the...Ch. 15 - Does the addition of a catalyst have any effects...Ch. 15 - 15.54 Consider the following equilibrium system...Ch. 15 - 15.55 Heating solid sodium bicarbonate in a closed...Ch. 15 - 15.56 Consider the following equilibrium...Ch. 15 - 15.57 What effect does an increase in pressure...Ch. 15 - Prob. 58QPCh. 15 - Consider the following equilibrium process: PCl 5...Ch. 15 - Consider the reaction: 2SO 2 ( g ) ⇄ 2 SO 3 ( g )...Ch. 15 - In the uncatalyzed reaction: N 2 O 4 ( g ) ⇄ 2 NO...Ch. 15 - 15.62 Consider the gas-phase reaction:
Predict...Ch. 15 - Consider the following equilibrium reaction in a...Ch. 15 - 15.64 The following diagrams show the reaction at...Ch. 15 - 15.65 The following diagrams show an equilibrium...Ch. 15 - 15.66 Consider the reaction . The first diagram...Ch. 15 - Prob. 67APCh. 15 - Consider the equilibrium system 3A → B . Sketch...Ch. 15 - Baking soda (sodium bicarbonate) undergoes thermal...Ch. 15 - Consider the following reaction at equilibrium: A...Ch. 15 - Prob. 71APCh. 15 - 15.72 Consider the following reacting...Ch. 15 - 15.73 At a certain temperature and a total...Ch. 15 - The decomposition of ammonium hydrogen sulfide: N...Ch. 15 - 15.75 Consider the following reaction at a certain...Ch. 15 - When heated, ammonium carbamate decomposes as...Ch. 15 - A mixture of 0 .47 mole of H2 and 3 .59 moles of...Ch. 15 - When heated at high temperatures, iodine vapor...Ch. 15 - 15.79 One mole of and three moles of are placed...Ch. 15 - At 1130°C , the equilibrium constant ( K c ) for...Ch. 15 - For the purpose of determining K p using Equation...Ch. 15 - The following diagram represents a gas-phase...Ch. 15 - 15.83 Consider the following reaction at
When...Ch. 15 - 15.84 A quantity of 0.20 mole of carbon dioxide...Ch. 15 - 15.85 When dissolved in water, glucose (com sugar)...Ch. 15 - 15 86 At room temperature, solid iodine is in...Ch. 15 - 15.87 A student placed a few ice cubes in a...Ch. 15 - 15.88 A mixture containing 3.9 moles of and 0.88...Ch. 15 - 15.89 The equilibrium constant for the...Ch. 15 - When heated, a gaseous compound A dissociates as...Ch. 15 - 15.91 When a gas was heated under atmospheric...Ch. 15 - The first diagram represents a system at...Ch. 15 - A sealed glass bulb contains a mixture of NO 2 and...Ch. 15 - At 20°C , the vapor pressure of water is 0.0231...Ch. 15 - A 2.50-mol sample of NOCl was initially in a...Ch. 15 - 15.96 About 75 percent of hydrogen for industrial...Ch. 15 - Water is a very weak electrolyte that undergoes...Ch. 15 - 15.98 Consider the following reaction, which takes...Ch. 15 - The equilibrium constant Kc for the reaction: 2NH...Ch. 15 - At 25°C, a mixture of NO 2 and N 2 O 4 gases are...Ch. 15 - 15.101 Consider the reaction between and in a...Ch. 15 - In 1899 the German chemist Ludwig Mond developed a...Ch. 15 - For which of the following reactions is K c equal...Ch. 15 - The equilibrium constant K p for the following...Ch. 15 - At 1024°C, , the pressure of oxygen gas from the...Ch. 15 - 15.06 The equilibrium constant for the following...Ch. 15 - 15.107 Industrially, sodium metal is obtained by...Ch. 15 - Consider the equilibrium reaction described in...Ch. 15 - The K p for the reaction: SO 2 Cl 2 ( g ) ⇄ SO 2 (...Ch. 15 - The "boat" form and the “chair" form of...Ch. 15 - A quantity of 6.75 g of SO 2 Cl 2 was placed in a...Ch. 15 - 15.112 Industrial production of ammonia from...Ch. 15 - 15.113 The equilibrium constant for the formation...Ch. 15 - Consider the reaction: 2NO( g )+ O 2 ( g ) ⇄ 2N O...Ch. 15 - The formation of SO 3 from SO 2 and O 2 is an...Ch. 15 - At 25°C , the equilibrium partial pressures of N O...Ch. 15 - 15.117 The vapor pressure of mercury is 0.0020...Ch. 15 - 15.118 Both ' and are important biological ions....Ch. 15 - Photosynthesis can be represented by: 6C O 2 ( g...Ch. 15 - Consider the decomposition of ammonium chloride at...Ch. 15 - 15.121 Eggshells are composed mostly of calcium...Ch. 15 - In the gas phase, nitrogen dioxide is actually a...Ch. 15 - Consider the potential-energy diagrams for two...Ch. 15 - Iodine is sparingly soluble in water but much more...Ch. 15 - The dependence of the equilibrium constant of a...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...Ch. 15 - Lime is used to prevent from escaping from the...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...Ch. 15 - Lime ( CaO ) is used to prevent SO 2 from escaping...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the following system, K=4.86105 at 298K: HClO(aq)+F(aq)ClO(aq)+HF(aq) Assuming that you start with equal concentrations of HClO and F, and that no ClO or HF is initially present, which of the following best describes the equilibrium system: a the forward reaction is favored at equilibrium; b the reverse reaction is favored at equilibrium; c appreciable quantities of all species are present at equilibrium?arrow_forwardThe equilibrium constant, Kp, for the following reaction is 0.215 at 673 K. NH4I(S) NH3(g) + HI(g) If an equilibrium mixture of the three compounds in a 7.23 L container at 673 K contains 2.31 mol of NH4I(s) and 0.237 mol of NH3(g), the partial pressure of HI(g) is atm. $ 4 Submit Answer F F4 % 5 T [Review Topics] [References] Use the References to access important values if needed for this question. Retry Entire Group 9 more group attempts remaining F5 G Cengage Learning | Cengage Technical Support A 6 MacBook Air ... Y H & 7 U J 8 ► 11 F8 ۔ 9 K Þ O ) –0 L F10 P 4) F11 Previous J + + 11 Next> Save and Exit 4) F12arrow_forwardWhy is an equilibrium constant unitless? Answer:arrow_forward
- 1:18 Question 2 of 16 Consider the following chemical reaction at equilibrium: 2NO₂(g) N₂O4(g) AH°rxn < 0 A) increase If the temperature is increased by 20 K while the volume is kept constant, how will Keq for the reaction change? B) decrease Submit C) stay the same Tap here or pull up for additional resourcesarrow_forward[References] For an equilibrium system involving gaseous substances, what effect, in general terms, is realized when the volume of the system is decreased? O The pressure is decreased and the reaction shifts in the direction that gives the larger number of gas molecules. O The pressure is increased and the reaction shifts in the direction that gives the smaller number of gas molecules. O The pressure is increased and the reaction shifts in the direction that gives the larger number of gas molecules. Submit Answer Try Another Version 1 item attempt remainingarrow_forward9. The equilibrium constant for the reaction 2 BrCl (g) → Br2 (g) + Cl2 (g) at 25°C is Keq = 0.0172. What is the equilibrium constant for the reaction Br2 (g) + Cl2 (g) →2 BrCl (g)? (а) -0.0172 (b) 0.131 (c) 7.62 (d) 58.1 (e) none of thesearrow_forward
- 2. At 450°C, the reaction below has a Keq value of 0.771. If 2.00 mol of both hydrogen and carbon dioxide are placed in a 4.00L container and allowed to react, what will be the equilibrium concentrations of all four gases? Equation: H, (g) CO, (g) + CO (g) + H,0 (g)arrow_forwardThe equilibrium constant, Ke, for the following reaction is 0.00650 at 298 K. 2NOBr(g) 2NO(g) + Br₂ (g) $ 4 Forms Office of t... KB Viewing Your Aca... Submit Answer If an equilibrium mixture of the three gases in a 17.8 L container at 298 K contains 0.378 mol of NOBr(g) and 0.461 mol of NO, the equilibrium concentration of Br₂ Is M. 000 000 F4 R F Show Hint % 5 Scholarship Ameri... [Review Topics] [References] Use the References to access important values if needed for this question. Retry Entire Group 9 more group attempts remaining T G Cengage Learning Cengage Technical Support ^ 6 Have Changes in... MacBook Air S Y H & 7 F7 U * 8 ►II F8 ( 9 K A F9 O -0 ) bio 1108 chat F10 SAVAGE X FENTY... Previous F11 +11 Next> Save and Exit F12 17arrow_forwardThe equilibrium constant for the following reaction is 6.50-10 at 298 2NOBrig)2NO(g) + Br₂(g) If an equilibrium mixture of the three gases in a 12.2 L container at 298K contains 0.343 mol of NOBrig) and 0.481 mol of NO, the equilibrium concentration of Brais [ Submit Answer 2 question attempts remainingarrow_forward
- For the following systems at equilibrium A: 2NOCI(g) B: H2(g) +I2(g) = 2NO(g)+Cl2 (g) = 2HI(g) classify these changes by their effect. Drag the appropriate items to their respective bins. • View Available Hint(s) Reset Help System A Increase container size System A Decrease container size System B Increase container size System B Decrease container size Leftward shift No shift Rightward shiftarrow_forwardAt 850 K, the equilibrium constant for the reaction 2 SO,(g) + 0,(g) = 2 SO, (g) is K = 15. If the given concentrations of the three gases are mixed, predict in which direction the net reaction will proceed toward equilibrium. Left No net reaction Right Answer Bank (SO,1 - 0.20 M 10,1-0.60 M (SO,) =0.60 M ISO,1-0.IN M 10,1-0.10M (SO,-050 M (0,-0.60 M ISO,I-0,15 M (S0,10.50 Marrow_forwardQ Search this course Reference Use the References to access importantv needed for this question. The equilibrium constant, K, for the following reaction is 1.20x102 at 500 K PCI5(g) PCl3(g) + Cl2(g) An equilibrium mixture of the three gases in a 1.00 L flask at 500 K contains 0.164 M PCI 4.43x10 M PCI3 and 4.43x10 M C2. What will be the concentrations of the three gases equilibrium has been reestablished, if 2.27x10 mol of Ch(g) is added to the flask? once [PCI5]= [PCI3]= М [Cl2] sturbing Equilibrium Concentrations (Qua... : This is group attempt 1 of 5 Next ack 1:33 PM 9/24/2019 up Xxarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY