
(a)
Interpretation: The products for radical chlorination and bromination of given compound are to be drawn. The compounds which form single constitutional isomer for both reactions are to be predicted. The true structure of a reactant for both reactions to form a single product is to be identified.
Concept introduction: Chlorination and bromination are radical substitution reaction. In the reaction of chlorination,
Answer to Problem 15.41P
The products of radical chlorination and bromination of given compound are,
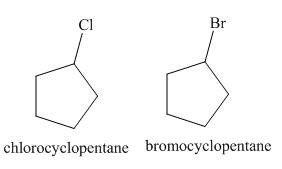
A single constitutional isomer is formed for both reactions.
Explanation of Solution
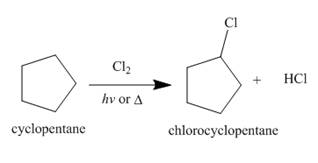
Figure 1
Alkanes undergo bromination through free radical mechanism when they are treated with
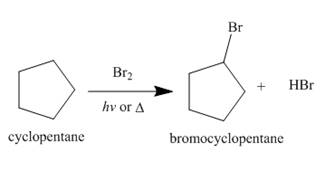
Figure 2
A single constitutional isomer is formed for both reaction because all hydrogen atoms of given compound are present in a same chemical environment. Therefore, to form a single product, the chemical environment of hydrogen atoms should be same.
The products of radical chlorination and bromination of given compound are shown in Figure 1 and Figure 2, respectively. A single constitutional isomer is formed for both reactions.
(b)
Interpretation: The products for radical chlorination and bromination of given compound are to be drawn. The compounds which form single constitutional isomer for both reactions are to be predicted. The true structure of a reactant for both reactions to form a single product is to be identified.
Concept introduction: Chlorination and bromination are radical substitution reaction. In the reaction of chlorination,
Answer to Problem 15.41P
The products of radical chlorination and bromination of given compound are,
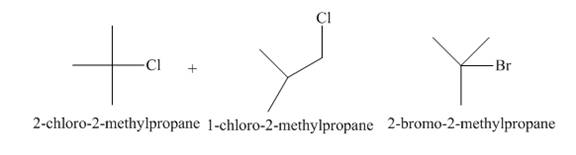
A single constitutional isomer is not formed for both reactions.
Explanation of Solution
Alkanes undergo chlorination when they are treated with
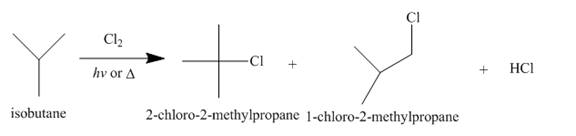
Figure 3
Alkanes undergo bromination by free radical mechanism when they are treated with
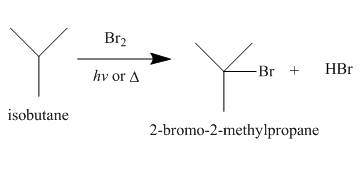
Figure 4
A single constitutional isomer is formed for bromination reaction but not for chlorination because bromine is more selective than chlorine.
The products of radical chlorination and bromination of given compound are shown in Figure 3 and Figure 4, respectively. A single constitutional isomer is not formed for both reactions.
(c)
Interpretation: The products for radical chlorination and bromination of given compound are to be drawn. The compounds which form single constitutional isomer for both reactions are to be predicted. The true structure of a reactant for both reactions to form a single product is to be identified.
Concept introduction: Chlorination and bromination are radical substitution reaction. In the reaction of chlorination,
Answer to Problem 15.41P
The products of radical chlorination and bromination of given compound are,
A single constitutional isomer is formed for both reactions.
Explanation of Solution
Alkanes undergo chlorination when they are treated with
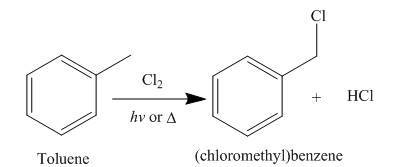
Figure 5
Alkanes undergo bromination when they are treated with
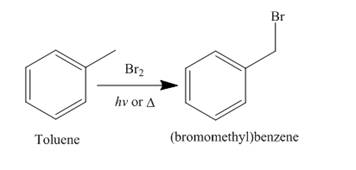
Figure 6
A single constitutional isomer is formed for both reaction because all hydrogen atoms of given compound are present in same chemical environment. Therefore, to form a single product, the chemical environment of hydrogen atoms should be same.
The products of radical chlorination and bromination of given compound are shown in Figure 5 and Figure 6, respectively. A single constitutional isomer is formed for both reactions.
(d)
Interpretation: The products for radical chlorination and bromination of given compound are to be drawn. The compounds which form single constitutional isomer for both reactions are to be predicted. The true structure of a reactant for both reactions to form a single product is to be identified.
Concept introduction: Chlorination and bromination are radical substitution reaction. In the reaction of chlorination,
Answer to Problem 15.41P
The products of radical chlorination and bromination of given compound are,
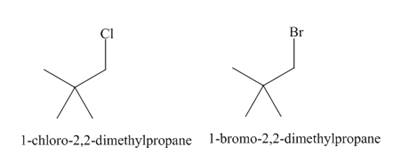
A single constitutional isomer is formed for both reactions.
Explanation of Solution
Alkanes undergo chlorination when they are treated with
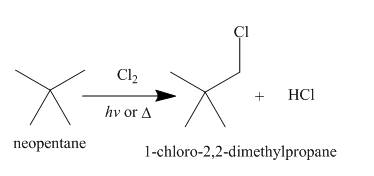
Figure 7
Alkanes undergo bromination when they are treated with
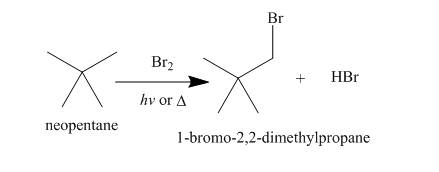
Figure 8
A single constitutional isomer is formed for both reaction because all hydrogen atoms of given compound are present in same chemical environment. Therefore, to form a single product, the chemical environment of hydrogen atoms should be same.
The products of radical chlorination and bromination of given compound are shown in Figure 7 and Figure 8, respectively. A single constitutional isomer is formed for both reactions.
(e)
Interpretation: The products for radical chlorination and bromination of given compound are to be drawn. The compounds which form single constitutional isomer for both reactions are to be predicted. The true structure of a reactant for both reactions to form a single product is to be identified.
Concept introduction: Chlorination and bromination are radical substitution reaction. In the reaction of chlorination,
Answer to Problem 15.41P
The products of radical chlorination and bromination of given compound are,
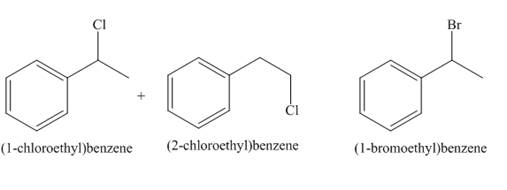
A single constitutional isomer is not formed for both reactions.
Explanation of Solution
Alkanes undergo chlorination when they are treated with
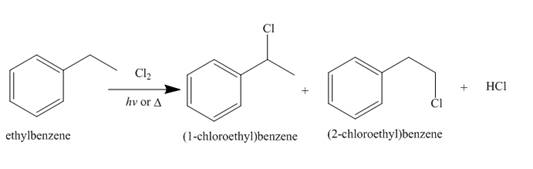
Figure 9
Alkanes undergo bromination by free radical mechanism when they are treated with
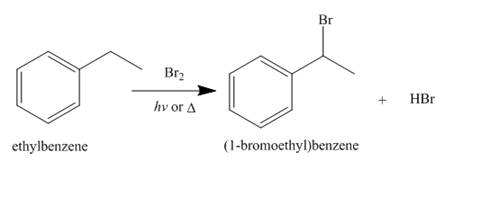
Figure 10
A single constitutional isomer is formed for bromination reaction but not for chlorination because bromine is more selective than chlorine.
The products of radical chlorination and bromination of given compound are shown in Figure 9 and Figure 10, respectively. A single constitutional isomer is not formed for both reactions.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry
- 1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forwardd) Explain why A is less reactive than B towards a nucleophilic attack. `H. A Вarrow_forwardidentify the reagents needed to come up with the compounds given. a. b. c. d. e. f. g. h. i.arrow_forward
- Electrophilic Addition Soubong neblA-aleid rose 9160910 31 babeen ene singo 14.43 Draw the products formed when each compound is treated with one equivalent of HBr. a. b. C.arrow_forwardWhich of the following compounds is aromatic? A. only A and B B. only A and C C. only B and C D.only Barrow_forward4.36 Give the IUPAC name for each compound. a. b. C. e. g. i. mর d. f. h.arrow_forward
- Synthesis 10.63 Devise a synthesis of each product from the given starting material. More than one step is required. a. b. d. e. Br Br OH OCH3 ta CI OHarrow_forwarda. b. C. XII. Provide proper IUPAC names for each compound below. Please PRINT! thearrow_forwardWhich heterocycles are aromatic? a. d. b.arrow_forward
- Which compound would be the least likely to react as a diene in a Diels- Alder Reaction? A. B. C. D.arrow_forward48. Which set of reagents would accomplish this conversion H. H. CH3OH with H* as a catalyst NaH followed by CH3OH CH3ONA followed CH3OH CH3ONA followed by CH3Br a. b. C. d.arrow_forwardAnswer the following questions about compounds A–D.a.How are the compounds in each pair related? Choose from constitutional isomers, stereoisomers, or identical molecules: A and B; A and C; B and D. b.Label each compound as a cis or trans isomer. c.Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents. d.Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





