
Concept explainers
Interpretation:
The product should be identified when two moles of methanol react with ethyl methyl
Concept Introduction:
Acid Catalyzed Hydration Reaction: The reaction involves breaking of
A hemiacetal or a hemiketal: addition of alcohol to an
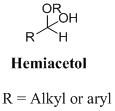
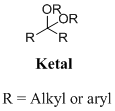
Ketal: A
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- In observing a Haworth or cyclohexane-chair representation of alpha-d-glucopyranose, the anomeric carbon can de best identified by: a) the carbon atom which is bonded to the most H atoms b) the carbon atom which is bonded to the least H atoms c) The carbon atom which has its OH group pointing down d) The carbon atom which is bonded to two oxygen atoms Cellulose differs from amylose in that: a) cellulose has 1-6 branches while amylose does not b) amylose has 1-6 branches while cellulose does not c) amylose has alpha glycosidic bonds while cellulose has beta glycosidic bonds d) cellulose contains sulfate while amylose does not. The glycosaminoglycan (mucopolysaccharide) which is not normally associated with joint tissue is: a) heparin b) keratan sulfate c) chondroitin sulfate d) hyaluronic acid In glycoproteins, which amino acid does not bond sugar molecules? a) Asn b) Ser c) Thr d) Gly Which feature do all lipid molecules share in common? a) fatty acid molecules…arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forwardFumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forward
- Draw the alcohol used to produce each of the following ketones. Include all hydrogen atoms.arrow_forwardWith the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forwardb) The above molecule may be further converted into a polymer with similar molecular structure to that of polyester. Describe the conditions of this process and write down the reaction for each step.arrow_forward
- Write the complete reaction for the isomerization of citrate. Name the enzyme that catalyses these reaction andcprovide the formulas of the reactants and the product of these reaction ?arrow_forwardGlycine hydrochloride (Cl− H3N+CH2COOH) is a diprotic acid that contains a carboxylic acid group and an ammonium group and is therefore called an amino acid. It is often used in biochemical buffers. Solve, In analogy with Figure , sketch the titration curve of this diprotic acid.arrow_forwardSalicylic acid (o-hydroxybenzoic acid) is used as starting material to prepare aspirin. Draw the structure of salicylic acid.arrow_forward
- Draw the predominant form of Gly-Gly when 2.0 moles of OH is added.arrow_forwardIn the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





