
Concept explainers
Interpretation:
Structure of the target compound should be drawn, when the starting alcohol molecule react with
Concept Introduction:
Acid Catalyzed Hydration Reaction: The reaction involves breaking of
Acetal formation: The acid catalysts is added to the reaction of an alcohol with an aldehyde or
A hemiacetal or a hemiketal: addition of alcohol to an aldehyde or ketone which produce hemi acetal.
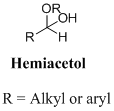
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Drawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardThis is the structure of 2,4-dimethyl-1-pentanol.arrow_forwardSalol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure of phenyl salicylate.arrow_forward
- When pentane is exposed to Br2 in the presence of light, a halogenation reaction occurs. Write the formulas of:(a) All possible products containing only one bromine(b) All possible products containing two bromines that are not on the same carbonarrow_forwardIdentify the functional groups in the following compoundsarrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forward
- With the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forwardThe metabolic intermediate acetyl phosphate is an anhydride formed from acetic acid and phosphoric acid. What is the structure of acetyl phosphate?arrow_forwardWhich of the alkenes is the most stable?arrow_forward
- Why do ketoses go dehydration reaction much faster than aldoses in Seliwanoff’s test when aldehyde reacts faster than ketoses? Kindly explain in a comprehensive manner.arrow_forwardWhat is the Lewis electron dot formula for formaldehyde (H2C=O)?arrow_forwardWhat functional groups are present in the peaks?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





