
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 64P
Give approximate wavenumbers for the major characteristic IR absorption bands that would be given by each of the following compounds:
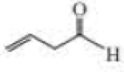
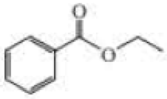
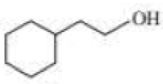
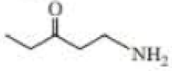
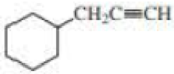
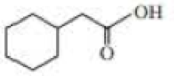
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
20) An unknown compound has the chemical formula CH₂O. It's IR spectrum and ¹H NMR
spectrum are shown below. Propose a structure for the unknown molecule. Calculate the
Degrees of Unsaturation (DU), assign any important peaks in the IR spectrum to vibrational
modes and briefly explain why the structure fits the 'H NMR spectrum that is provided
Degrees of unsaturation (show work below):
442
Du= (2(3)+2)-6
2
-8-16 - 2²/2/2 = 1
3437
1739
Amply
0
Vibrational Assignments of IR Peaks
Wavenumber (cm-¹) Vibrational Assignment
0-4 stretch
6=0 strekh
Peak 1:
9.79 ppm
14
Singlet
3
8
Brief rationalization for proposed structure: (explain why DU, IR, and
NMR are consistent with proposed structure)
ppm
Peak 2
2.45 ppm
2H
Quartet
Peak 3
1.11 ppm
3H
Triplet
D
Proposed structure of unknown:
Give approximate wavenumbers for the major characteristic IR absorption bands that would be given by each of the following compounds
The IR spectrum below belongs to an organic compound A having one nitrogen atom.
Determine which structures that fits to the IR spectrum. Justify your answer by providing two (2)
IR absorption peaks with their wavenumber.
CN
NH2
Ahsorbance / 4
50
100-
4000
3000
2500
2000
1500
1000
500
Wavenumbers / cm
Chapter 14 Solutions
Organic Chemistry
Ch. 14.1 - Which of the following fragments produced in a...Ch. 14.2 - What distinguishes the mass spectrum of...Ch. 14.2 - What is the most likely m/z value for the base...Ch. 14.3 - Prob. 5PCh. 14.3 - If a compound has a molecular ion with an...Ch. 14.3 - a. Suggest possible molecular formulas for a...Ch. 14.3 - Identify the hydrocarbon that has a molecular ion...Ch. 14.4 - Predict the relative intensities of the molecular...Ch. 14.5 - Which molecular formula has an exact molecular...Ch. 14.5 - Prob. 11P
Ch. 14.6 - Sketch the mass spectrum expected for...Ch. 14.6 - The mass spectra of 1-methoxybutane,...Ch. 14.6 - Prob. 14PCh. 14.6 - Identify the ketones responsible for the mass...Ch. 14.6 - Prob. 16PCh. 14.6 - Using curved arrows, show the principal fragments...Ch. 14.6 - The reaction of (Z)-2-pentene with water and a...Ch. 14.9 - Prob. 19PCh. 14.9 - Prob. 20PCh. 14.9 - Prob. 21PCh. 14.13 - Prob. 22PCh. 14.14 - Which occur at a larger wavenumber: a. the C O...Ch. 14.14 - Prob. 24PCh. 14.14 - Prob. 25PCh. 14.14 - Rank the following compounds from highest...Ch. 14.14 - Which shows an O H stretch at a larger...Ch. 14.15 - Prob. 28PCh. 14.15 - a. An oxygen-containing compound shows an...Ch. 14.15 - Prob. 30PCh. 14.15 - For each of the following pair of compounds, name...Ch. 14.16 - Which of the following compounds has a vibration...Ch. 14.16 - Prob. 33PCh. 14.17 - A compound with molecular formula C4H6O gives the...Ch. 14.19 - Prob. 35PCh. 14.19 - Prob. 36PCh. 14.20 - Predict the max of the following compound:Ch. 14.20 - Prob. 38PCh. 14.21 - a. At pH = 7 one of the ions shown here is purple...Ch. 14.21 - Prob. 40PCh. 14.22 - Prob. 41PCh. 14.22 - Prob. 42PCh. 14 - In the mass spectrum of the following compounds,...Ch. 14 - Prob. 44PCh. 14 - For each of the following pairs of compounds,...Ch. 14 - Draw structures for a saturated hydrocarbon that...Ch. 14 - a. How could you use IR spectroscopy to determine...Ch. 14 - Assuming that the force constant is approximately...Ch. 14 - In the following boxes, list the types of bonds...Ch. 14 - A mass spectrum shows significant peaks at m/z. =...Ch. 14 - Prob. 51PCh. 14 - Prob. 52PCh. 14 - Prob. 53PCh. 14 - How can you use UV spectroscopy to distinguish...Ch. 14 - Rank the following compounds from highest...Ch. 14 - Rank the following compounds from highest...Ch. 14 - What peaks in their mass spectra can be used to...Ch. 14 - Each of the IR spectra shown below is accompanied...Ch. 14 - Prob. 59PCh. 14 - Prob. 60PCh. 14 - How can IR spectroscopy distinguish between...Ch. 14 - 62. Draw the structure of a carboxylic acid that...Ch. 14 - Prob. 63PCh. 14 - Give approximate wavenumbers for the major...Ch. 14 - Prob. 65PCh. 14 - Prob. 66PCh. 14 - Prob. 67PCh. 14 - The IR spectrum of a compound with molecular...Ch. 14 - Which one of the following live compounds produced...Ch. 14 - Prob. 70PCh. 14 - Phenolphthalein is an acid-base indicator. In...Ch. 14 - Which one of the following five compounds produced...Ch. 14 - The IR and mass spectra for three different...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many 13C peaks should be seen in the broad-band decoupled spectrum of the structure at the right?arrow_forwardThe IR spectra of two isomers are given below. Identify which IR spectra belong to isopropylbenzene and propylbenzene, respectively. How we decide them?arrow_forwardIdentify the important absorption peak and its description in the IR spectra.arrow_forward
- Explain the following trend in the absorptions corresponding to the most energetic C–H stretching mode observed in the IR spectra of the following molecules.arrow_forwardWhy are n-π* transitions generally of low intensities/low absorptions? What photon energy and wavelength would you need to send on a compound having an energy gap (Eg) of 2.7 eV? Detail your answer with appropriate information. Using molecular orbital theory/concept (draw the important molecular orbitals), explain the difference in absorption for the phenolphthalein in neutral medium or in basic medium, both for the n-π* and the π-π* transition.arrow_forwardA peak at a wavenumber of 1656 cm-1 is observed for caffeine. Calculate the wavelength and frequency of this radiation and the energy change due to this absorption.arrow_forward
- What is the structure of the unknown? Determine it by analyzing the spectroscopic data given in IR, MS, 1H NMR, 13C NMR (Broadband decoupled with carbon types) and fully substantiate your answer with key supporting data from each spectrum.arrow_forwardAssign bond types that correspond to the absorption in the IR spectrum of unknown X.arrow_forwardThe spectrophotometer is used to measure absorbance experimentally. Briefly, explain how this is accomplished.arrow_forward
- 3604 84 3352 81 3007 11 12 3063 64 3040 72 3429 12 3006 68 2967 77 2925 79 2667 04 1606 4 1546 GE 1599 21 1603 41 Anaya 21) An unknown compound has the chemical formula CgH8O. It's IR spectrum and ¹H NMR spectrum are shown below. Propose a structure for the unknown molecule. Calculate the modes and briefly explain why the structure fits the ¹H NMR spectrum that is provided. Degrees of Unsaturation (DU), assign any important peaks in the IR spectrum to vibrational 5 Degrees of unsaturation (show work below): 16+2. DU = (218) 727-8-18-8 - 10/=5 OH 1645 61 1492 1 1450 26 1430 62 1360 13 1315 82 1303 53 2000 4267 6 1161 St LIED 74 1103 79 1079 62 1025 50 1001 74 966 37 928 72 761 15 731 79 691 14 618 81 688 17 2 1500 2 1000 Vibrational Assignments of IR Peaks Wavenumber (cm-¹) 73355 13087-3005 2957-2857 Vibrational Assignment 0-H Stretch Ct Stretch 2-1 Stretch (=C 9 Name: Region from 8.0-7.3 ppm Zoomed in Peak 1: 7.96 ppm 2H Doublet HR201102501NS Peak 2: 7.56 ppm 1H Triplet TTTTTTTTTTTE…arrow_forward12) What is a reasonable structure for a molecule with the formula C6H7N and the following IR spectrum? Be sure to justify your structure by indicating important peaks along with the intensities and breadth of those peaks that helped in your determination. ww LOD D 4000 3000 2000 HAVENUMBERI 1500 1000 500arrow_forwardWhich of the following molecules would be expected to have absorbance in the IR range from an asymmetric stretching vibration? CO2 CF4 CCl4 NO2 All of the above.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY