
Concept explainers
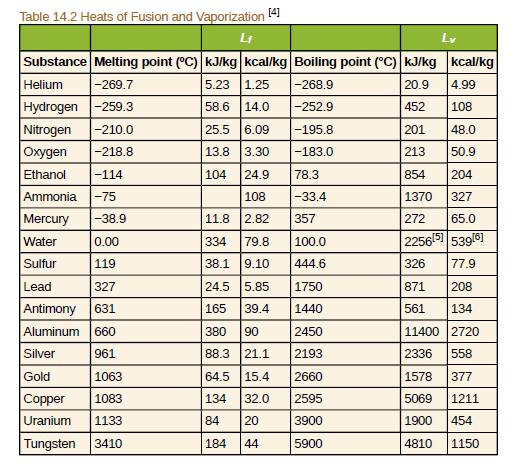
In a physics classroom demonstration, an instructor inflates a balloon by mouth and then cools it in liquid nitrogen. When cold, the shrunken balloon has a small amount of light blue liquid in it, as well as some snow-like crystals. As it warms up, the liquid boils, and part of me crystals sublimate, with some crystals lingering for a while and then producing a liquid. Identify the blue liquid and the two solids in the cold balloon. Justify your identifications using data from Table 14.2.
Trending nowThis is a popular solution!

Chapter 14 Solutions
College Physics
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Lecture- Tutorials for Introductory Astronomy
College Physics: A Strategic Approach (4th Edition)
Conceptual Physics (12th Edition)
University Physics Volume 2
Sears And Zemansky's University Physics With Modern Physics
- A liquid with a coefficient of volume expansion just fills a spherical shell of volume V(Fig. P19.51). The shell and the open capillary of area A projecting from the top of the sphere are made of a material with an average coefficient of linear expansion . The liquid is free to expand into the capillary. Assuming the temperature increases by T find the distance h the liquid rises in the capillary.arrow_forwardThe surface tension and viscosity of water at several different temperatures are given in this table. (a) As temperature increases, what happens to the surface tension of water? Explain why this occurs, in terms of molecular interactions and the effect of changing temperature.(b) As temperature increases, what happens to the viscosity of water? Explain why this occurs, in terms of molecular interactions and the effect of changing temperature.arrow_forwardThe combined gas law PV=nRT is a very useful equation. From the equation one can see the relationships between the variables. Scenario: I just climbed to the top of mount Everest and it is a beautiful day. For a celebration I want to eat a dehydrated meal with raw chicken. This is safe to do so? Questions to think about: What pressure is present on the top of Mt. Everest? Greater or less than the pressure at sea level? How does that relate to temperature? What temperature will water boil at the top of Mt. Everest? Will that temperature be hot enough to cook the raw chicken?arrow_forward
- 1. What would happen to the water level in a glass if the ice cube floating in a glass of water will be completely melted? 2. A. Why does a balloon filled with helium gas rise? B. Is there a lower limit on how much helium gas it must contain before it begins to rise?arrow_forwardI put air in the tires of my car during the summer, while it was 28.8 °C outside (about 83.8 °F). My tires say to fill them to a pressure of 203,000 Pa. Boltzmann's constant is 1.38 * 10 ^ - 23 * J / K A) If my tires have a volume of 0.0100 m³, how many air molecules are in the tire? N = molecules B) These tires go all summer without losing any air, however come fall the temperature drops down to 8.65 °C (about 47.6 °F). Assuming the volume doesn't change (because it doesn't) what is the new pressure in my tires? P=arrow_forwardThe A and B tanks given in the figure are separated from each other by the closed valve between them. The water vapor in tank A has a volume of 0.2 m, a temperature of 150 ° C and a pressure of 100 kPa. Tank B is filled with saturated water vapor at a volume of 0.3 m and a pressure of 300 kPa. Then, the valve between the tanks is opened and the final pressure is 175 kPa as a result of the mixing of the fluids in the tanks with each other and the transfer of sufficient . According to this; a) Determine the temperature (° C) and phase of the fluid in the final state. b) The amount of heat transfer at the end of the process (kJ)arrow_forward
- The water is smooth and continuous. Water droplets cling to your skin, and your skin temperature is cooler? Explain these observations based on the properties of waterarrow_forward1. The pressure gauge shown in Figure 1 consists of a spring inside vacuum chamber. The chamber is isolated from the a environment by a piston, which is free to move up or down. Ambient pressure exerts a force on the piston, which compresses the spring. The piston has a diameter of 30 mm. At sea level, where ambient pressure is 1 atm, the spring is compressed by distance xo = 14 mm relative to its relaxed length. Suppose the pressure gauge is lowered into water at a lake. At depth D, the spring is compressed by distance x, = 34 mm relative to its relaxed length. Calculate D. Show and explain your work. Vacuum Figure 1arrow_forwardWhich diagram(s) O 1. II and III only 2. All of these О3. I only O 4. III only I II O 5. I and III only O 6. I and II only O 7. II only 8. None of these III best characterize(s) a pure substance that is a compound?arrow_forward
- The column of mercury in a barometer (see Figure below) has a height of 0.760 m when the pressure is one atmosphere and the temperature is 0.0 °C. Ignoring any change in the glass containing the mercury, what will be the height of the mercury column for the same one atmosphere of pressure when the temperature rises to 33.1 °C on a hot day? Hint: The pressure in the barometer is Pressure pgh, and the density p changes when the temperature changes. Number i Units ……….…………ມາ WIII | Empty, except for a negligible amount of mercury vapor (P₁ = 0 Pa) B (P2= Atmospheric pressure)arrow_forwardThe figure below shows a section of hot-water pipe consisting of three parts: a straight, horizontal piece h = 28.0 cm long, a straight vertical piece ℓ = 110 cm long, and an elbow joint that allows the two pipe sections to meet at a right angle. An illustration shows the inside of a wall where an L-shaped water pipe goes through it. The vertical part from the top of the wall to the elbow is labeled ℓ. The horizontal part from the elbow to the beam in the wall is labeled h. A stud and a second-story floorboard hold the ends of this section of pipe stationary. The pipe is made of a copper alloy with a coefficient of linear expansion of 1.7 ✕ 10−5(°C−1). When the water flow is turned on, the temperature of the pipe rises from 18.0°C to 42.5°C. What is the magnitude and direction of the displacement of the pipe elbow from its original location once the pipe reaches the higher temperature? (Give the magnitude in mm and the direction in degrees below the +x-axis, which is parallel to the…arrow_forwardA vertical tube that is closed at the upper end and open at the lower end contains an air pocket. The open end of the tube is under the water of a lake, as shown in the figure. When the lower end of the tube is just under the surface of the lake, where the temperature is 37degC and the pressure is 1.0×105Pa, the air pocket occupies a volume of 630cm3. Suppose now that the lower end of the tube is at a depth of 46m in the lake, where the temperature is 17.0degC. What is the volume of the air pocket under these conditions?arrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

