
Basic Chemistry
6th Edition
ISBN: 9780134878119
Author: Timberlake, Karen C. , William
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 57UTC
Interpretation Introduction
Interpretation:
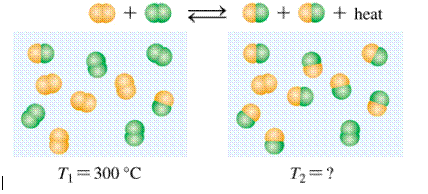
‘Temperature of product is lower or greater than reactant’ should be identified.
Concept introduction:
Equilibrium is the condition at which the rate of formation of product is equal to rate of disappearance of reactant.
Exothermic reaction is the reaction which liberates energy and the temperature of the product is lower than reactants to proceed the reaction.
Given:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(LO 7.1.1, 7.1.2, 7.2.1, 7.2.2) A small home might use about 6.50 moles of methane,
CH4, per day in moderately cold weather for heating. How many moles of water will
be produced when combusting 6.50 moles of methane?
CH4 +
CO₂ +
2.17
3.25
4.33
6.5
9.75
13.0
19.5
0₂ →
111-
H₂O (unbalanced)
Barium Hydroxide has a solubility of 4.68 g Ba(OH)2 in 100. g H2O at 25°C. Determine if each of the following
solutions will be saturated or unsaturated at 25°C: (9.3)
a) adding 32 g of Ba(OH)2 to 990 g of H2O
b) adding 7.0 g of Ba(OH)2 to 125 g of H2O
c) adding 22 g of Ba(OH)2 to 350. g of H2O
(Q28) How many grams of phosphine (PH3) gas can form when an excess of solid phosphorus (P4)
reacts in a sealed chamber with 56.2 liters of hydrogen (H2) gas at STP? (3 sf)
Chapter 13 Solutions
Basic Chemistry
Ch. 13.1 - Prob. 1PPCh. 13.1 - Prob. 2PPCh. 13.1 - In the following reaction, what happens to the...Ch. 13.1 - Prob. 4PPCh. 13.1 - Prob. 5PPCh. 13.1 - Prob. 6PPCh. 13.2 - What is meant by the term reversible reaction?Ch. 13.2 - Prob. 8PPCh. 13.2 - Which of the following are at equilibrium? a. The...Ch. 13.2 - Which of the following are not at equilibrium? a....
Ch. 13.2 - 13.11 The following diagrams show the chemical...Ch. 13.2 - Prob. 12PPCh. 13.3 - Write the equilibrium expression for each of the...Ch. 13.3 - Prob. 14PPCh. 13.3 - Prob. 15PPCh. 13.3 - Prob. 16PPCh. 13.3 - What is the numerical value of Kc for the...Ch. 13.3 - What is the numerical value of Kc for the...Ch. 13.3 - What is the numerical value of Kc for the...Ch. 13.3 - What is the numerical value of Kc for the...Ch. 13.3 - Prob. 21PPCh. 13.3 - Identify each of the following as a homogeneous or...Ch. 13.3 - Prob. 23PPCh. 13.3 - Prob. 24PPCh. 13.3 - Prob. 25PPCh. 13.3 - What is the numerical value of Kc for the...Ch. 13.4 - Prob. 27PPCh. 13.4 - Prob. 28PPCh. 13.4 - Indicate whether each of the following equilibrium...Ch. 13.4 - Indicale whether each of the following equilibrium...Ch. 13.4 - Prob. 31PPCh. 13.4 - The numerical value of the equilibrium constant,...Ch. 13.4 - Prob. 33PPCh. 13.4 - The numerical value of the equilibrium constant,...Ch. 13.5 - In the lower atmosphere, oxygen is converted to...Ch. 13.5 - Prob. 36PPCh. 13.5 - Hydrogen chloride can be made by reacting hydrogen...Ch. 13.5 - When heated, carbon monoxide reacts with water to...Ch. 13.5 - Use the following equation for the equilibrium of...Ch. 13.5 - Use the following equation for the equilibrium of...Ch. 13.5 - Prob. 41PPCh. 13.5 - Prob. 42PPCh. 13.6 - For each of the following slightly soluble ionic...Ch. 13.6 - For each of the following slightly soluble ionic...Ch. 13.6 - Prob. 45PPCh. 13.6 - Prob. 46PPCh. 13.6 - A saturated solution of silver carbonate, Ag2CO3 ,...Ch. 13.6 - Prob. 48PPCh. 13.6 - Calculate the molar solubility, S , of CuI if it...Ch. 13.6 - Calculate the molar solubility, S , of SnS if it...Ch. 13.6 - The CO2 level in the atmosphere has increased over...Ch. 13.6 - Prob. 52PPCh. 13 - Write the equilibrium expression for each of the...Ch. 13 - Write the equilibrium expression for each of the...Ch. 13 - Prob. 55UTCCh. 13 - Would the equilibrium constant, Ke , for the...Ch. 13 - Prob. 57UTCCh. 13 - Prob. 58UTCCh. 13 - Prob. 59APPCh. 13 - Prob. 60APPCh. 13 - For each of the following reactions, indicate if...Ch. 13 - For each of the following reactions, indicate if...Ch. 13 - Consider the reaction: (13.3) 2NH3(g)N2(g)+3H2(g)...Ch. 13 - Prob. 64APPCh. 13 - Prob. 65APPCh. 13 - Prob. 66APPCh. 13 - Prob. 67APPCh. 13 - According to Le Châtelier's principle, does the...Ch. 13 - Prob. 69APPCh. 13 - Prob. 70APPCh. 13 - The numerical value of the equilibrium constant,...Ch. 13 - The numerical value of the equilibrium constant,...Ch. 13 - For each of the following slightly soluble ionic...Ch. 13 - For each of the following slightly soluble ionic...Ch. 13 - Prob. 75APPCh. 13 - Prob. 76APPCh. 13 - Prob. 77APPCh. 13 - Prob. 78APPCh. 13 - What is the molar solubility, S , of CdS if it has...Ch. 13 - Prob. 80APPCh. 13 - Prob. 81CPCh. 13 - Prob. 82CPCh. 13 - Prob. 83CPCh. 13 - Indicate how each of the following will affect the...Ch. 13 - Prob. 85CPCh. 13 - Prob. 86CPCh. 13 - Prob. 87CPCh. 13 - Prob. 88CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 9.116 Lithium chloride has a solubility of 55 g of LiCl in 100. g of H,O at 25 °C. Determine if each of the following mixtures forms an unsaturated or saturated solution at 25 °C: (9.3) a. adding 10 g of LiCl to 15 g of H;O b. adding 25 g of LİCI to 50. g of H,0 c. adding 75 g of LiCl to 150. g of H,O ne on nearrow_forward(5.8)Which of the following reactions will form a gaseous product? O H₂CO3(aq) + Pb(NO3)2(aq) O NaOH(aq) + HNO3(aq) O None of these O Na₂SO3(aq) + H₂SO4(aq) ◄ Previousarrow_forwardshould react most rapidly with Cl2 and AICI. (9.28.h) should have the highest boiling point, 7. Of the compounds shown below, B A. D. 8. Of the compounds shown below, NO2 HO, is not aromatic. (9.1 lg,i) 9. Of the compounds shown below, only - D Z-I Uarrow_forward
- 10.105 Complete and balance each of the following: (10.7) a. ZnCO3(s) + H,SO,(aq) b. Al(s) + HBr(aq)arrow_forward6.(0-2) Black powder was invented in China in the 9th century. It was used as an explosive with propelling properties. Today it is used in fireworks, model rocket engines, and replica black powder weapons. Black powder consists of potassium nitrate (potassium nitrate, KN03), charcoal and sulfur. A variety of chemical reactions take place when this mixture is exploded. One of these is shown below. 2KNO, +S+ 30 → K, S+N, ↑+3C0, 1 Knowing that carbon makes up 15% of the black powder, calculate the total volume of gases (under normal conditions) released when 0.1 kg of black powder explodes. Assume that only the chemical reaction given above occurs.arrow_forwardOne of the reactions in the industrial production ofnitric acid involves the production of nitric oxide: 4 NH3(g) 1 5 O2(g) → 4 NO(g) 1 6 H2O(g) (7.4) T/I a) If 4500 kg of ammonia, NH3(g), react with7500 kg of O2, what mass of NO will form? (b) What mass of the excess reagent will remain?arrow_forward
- 9:30 P CHEM124 CHEMISTRY F. https://coetbschemfabon.neolm. (6.11) Calcium hydride, CaH2 (s); reacts with water to yield H2 and Ca(OH)2 (s). (g) (a) Write the balanced equation for this reaction. (b) How many grams of CaH2 (s) is required to prepare 3.00 L of H2 (g) at STP? Atomic mass: Ca 40.078 H 1.008 O 16.00 Analysis (Given): StandardP = (supply with unit) Standard T = degree Celcius Answer: (a) Balanced equation: CAH2(s) + H2O(g) H2(g) Ca(ОН)2 (s) mol H2 formed = (3 sig fig) (based on ideal gas equation: PV = nRT) mass CaH2 required = g CaH2 (3 sig fig)arrow_forward11.114 2.00 L of N,, at 25 °C and 1.08 atm, is mixed with 4.00 I of O2, at 25 °C and 0.118 atm, and the mixture is allowed to react. How much NO, in grams, is produced? (9.2, 9.3, 11.7, 11.8) N2(g) + O2(8) → 2NO(8)arrow_forwardFor the following reactions at equilibrium, identify which between the reactants and products is dominant. (ONLY 3 AND 4)arrow_forward
- Fill in the blanks: (CH3),COK THE Br The reaction will most likely proceed via the mechanism because of the following reasons: 1. 3. 2.arrow_forward(6.5)What is the molar volume of argon gas at STP? O 15.3 L O 22.4 L O 4.00 L O 1.00 Larrow_forwardJania.20 Chapter 3 3.100 When 1.0 9 of gasoline burns, it releases gasoline 0.749/ML SI (3.4.3.6) A) HOw many megajoules arc released srs 1.0 gai ot gasoline burns? Whenarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY