
Interpretation: The product formed after reaction of cyclohexane in the presence of
Concept introduction:
Molecules that have one unpaired electron are called free radicals.
Answer to Problem 1PP
Solution:

Explanation of Solution
Given information:
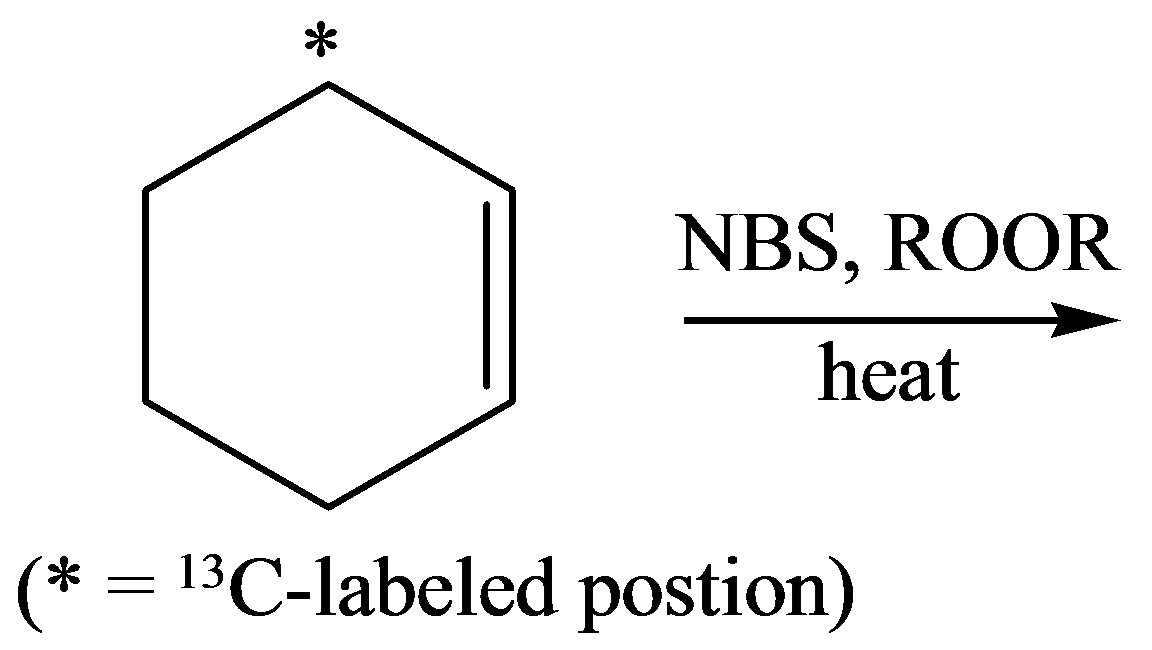
The reaction in the presence of
Allylic bromination is a free radical substitution reaction in which removal of hydrogen atom on carbon adjacent to a double bond with bromine.
Free radical substitution reaction takes place in the presence of
The structure of 3-bromocyclohexence is as follows:

The product formed after free radical substitution is 3-bromocyclohexence.
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY-ETEXT REG ACCESS
- NaOCH3 (S)-1-chloro-1,2-diphenylethane مين من cis-1,2-diphenylethene trans-1,2-diphenylethene + NaCl + HOCH 3 (a) Disregarding stereochemistry for the moment, provide a curved arrow mechanism for the transformation of the starting alkyl halide to one of the products.arrow_forwardmost of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the addition to phenanthrene gives a mixture of synand anti stereochemistry. When the product from (c) is heated, HBr is evolved and 9-bromophenanthrene results.Propose a mechanism for this dehydrohalogenation.arrow_forwardolo (S)-1-chloro-1,2-diphenylethane NaOCH3 + NaCl + HOCH3 cis-1,2-diphenylethene trans-1,2-diphenylethene (a) Disregarding stereochemistry for the moment, provide a curved arrow mechanism for the transformation of the starting alkyl halide to one of the products.arrow_forward
- Q12. (1-bromoethyl)benzene 1 udergoes an elimination following an E1 mechanism. Fill in the following synthetic scheme by drawing the structure of intermediate 2 and product 3. The chemical formula of product 3 is provided as guidance. CH; RDS Br C3Hg E1 2 3arrow_forwardProvide the major products (write “no reaction” if you think so) for the following reactions with correct stereochemistry.arrow_forward5. Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? Which product will be the major product in each case? çI CHa CH3CHCH2-C-CHCH3 Br ÇH3 CH3CH2CHCHCH3 (a) (b) ÇH3 (c) Br -CHCH3 ČH3arrow_forward
- Complete the following reaction scheme. Give all product(s) and indicate major or minor and any relevant stereochemistry (g) 1) BH3. THF 2) H202, OH (h) 1) Hg(OАC), H,о 2) NABH4arrow_forwardHow many alkenes yield 2,2,3,4,4−pentamethylpentane on catalytic hydrogenation?arrow_forward1. Bicyclic keto compounds such as camphor react predictably with nucleophiles according to steric influences; this effect has been termed steric approach control. In contrast, the reaction of nucleophiles with simple acyclic and monocyclic keto compounds are influenced primarily by thermodynamic factors; this effect has been termed product development control. Given the following reaction, predict the identity of the major product and provide a rationale for your selection. ОН NABH4 t-Bu. t-Bu t-Bu. ELOH 1 2arrow_forward
- Compounds X and Y are both C7H15Cl products formed in the radical chlorination of 2,4-dimethylpentane. Base-promoted E2 elimination of X and Y gives, in each case, a single C7H₁4 alkene. Both X and Y undergo an SN2 reaction with sodium iodide in acetone solution to give C7H15l products; in this reaction Y reacts faster than X. What is the structure of X? • Do not use stereobonds in your answer. • In cases where there is more than one possible structure for each molecule, just give one for each. . Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. наarrow_forwardShow how the following ethers might be synthesized using (1) alkoxymercuration–demercuration and (2) the Williamson synthesis. (When one of these methods cannot beused for the given ether, point out why it will not work.)(a) 2-methoxybutanearrow_forwardAlthough there are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

